Question
1. The atomic numbers of elements C and D are 19 and 9 respectively. State and explain the electrical conductivity of the compound CD in:
1. The atomic numbers of elements C and D are 19 and 9 respectively. State and explain the electrical conductivity of the compound CD in:
(a)Solid state(1½marks)
(b)aqueous state.
2. (a)Distinguish between a covalent bond and a co-ordinate bond.
(b)Draw a diagram to show bonding in an ammonium ion.
3. (a)Explain why the metals magnesium and aluminium are good conductors of electricity.(1 mark)
(b)Other than cost, give two reasons why aluminium is used for making electric cables while magnesium is not.
4. Both chlorine and iodine are halogens.
(a)What are halogens?
(b)In terms of structure and bonding, explain why the boiling point of chlorine is lower than that of iodine.
5. The diagram below is a section of a model of the structure of element T.
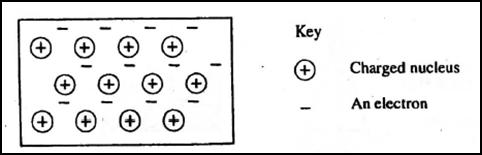 (a)State the type of bonding that exists in T.
(a)State the type of bonding that exists in T.
(b)In which group of the period table does element T belong? Give a reason.
Key + Charged nucleus An electron
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started

