Answered step by step
Verified Expert Solution
Question
1 Approved Answer
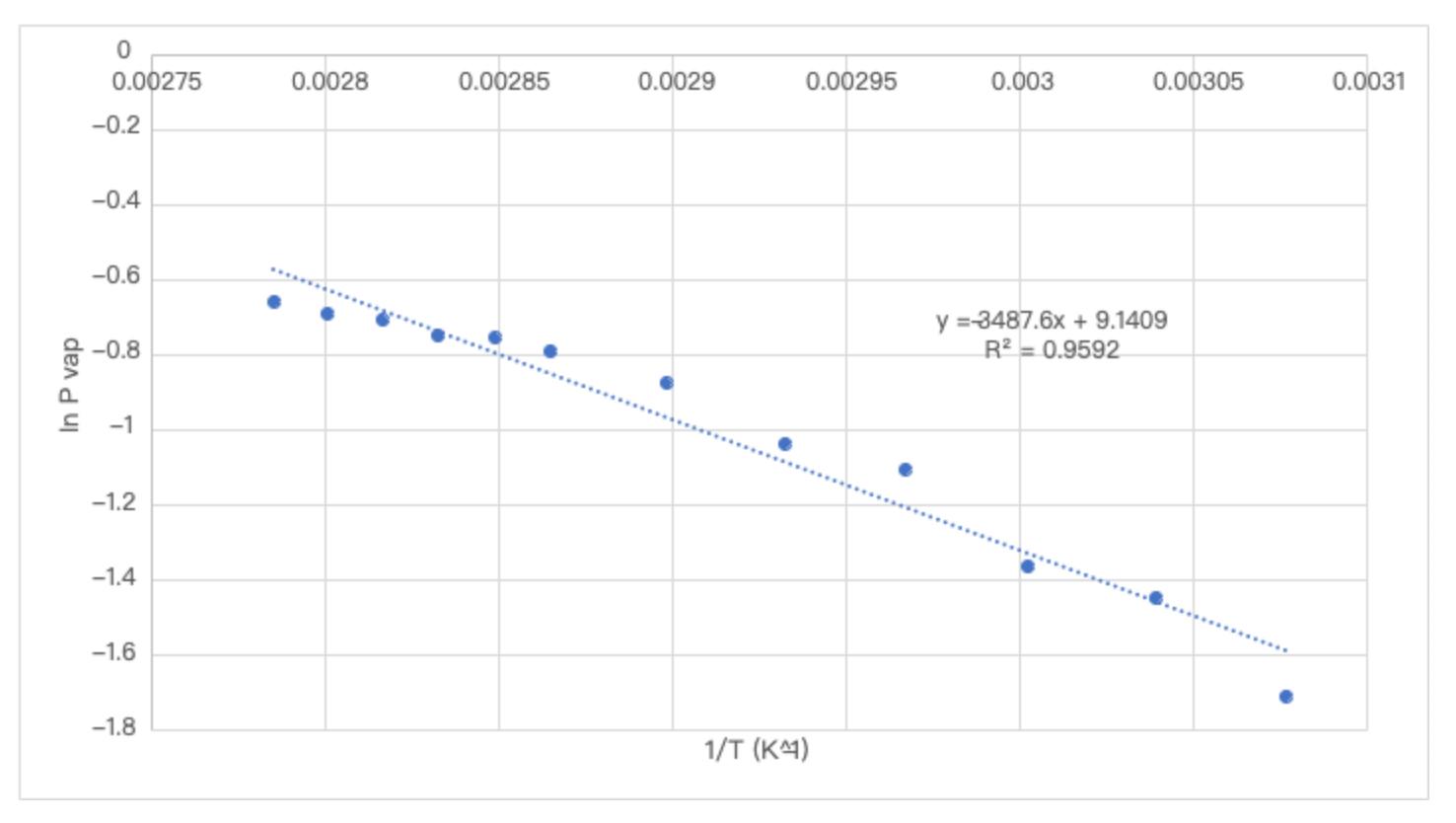
1 . Would you expect the Hvap for pentane ( CH 3 CH 2 CH 2 CH 2 CH 3 ) to be higher or
Would you expect the Hvap for pentane CHCHCHCHCH to be higher or lower than that of water? Why?
Would you expect the addition of a small amount of a salt the water to increase or decrease the vapor pressure readings at each temperature? If you are unsure, think about whether the solution would have higher or lower IMFs than the pure water, and what that would do to the Hvap for the solution compared to pure water. Other things to think about are Henrys law and the effect of a solute on boiling point. How might dirty glassware affect your results? talk about this as a source of error
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


