Answered step by step
Verified Expert Solution
Question
1 Approved Answer
2 questions Enter your answer in the provided box. How many grams of phosphine (PH3) can form when 18.7g of phosphorus and 93.1L of hydroge
2 questions 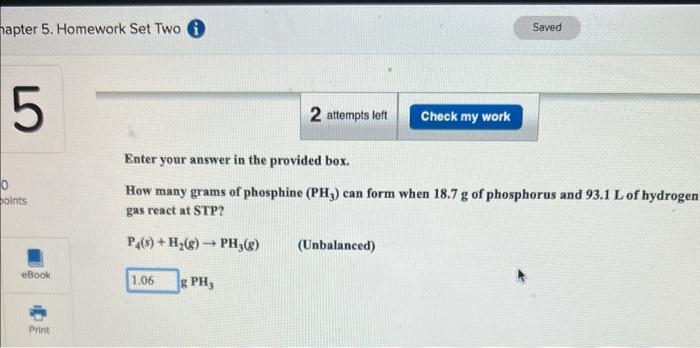
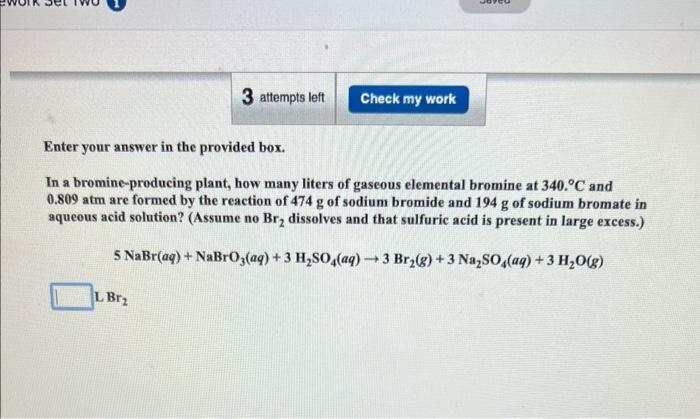
Enter your answer in the provided box. How many grams of phosphine (PH3) can form when 18.7g of phosphorus and 93.1L of hydroge gas react at STP? P4(s)+H2(g)PH3(g) (Unbalanced) Enter your answer in the provided box. In a bromine-producing plant, how many liters of gaseous elemental bromine at 3400C and 0.809atm are formed by the reaction of 474g of sodium bromide and 194g of sodium bromate in aqueous acid solution? (Assume no Br2 dissolves and that sulfuric acid is present in large excess.) 5NaBr(aq)+NaBrO3(aq)+3H2SO4(aq)3Br2(g)+3Na2SO4(aq)+3H2O(g) 

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


