Answered step by step
Verified Expert Solution
Question
1 Approved Answer
3 A piston cylinder system is initially split into two chambers using a fixed, insulated membrane. Chamber A contains 1.5 kg of saturated vapor ammonia
3 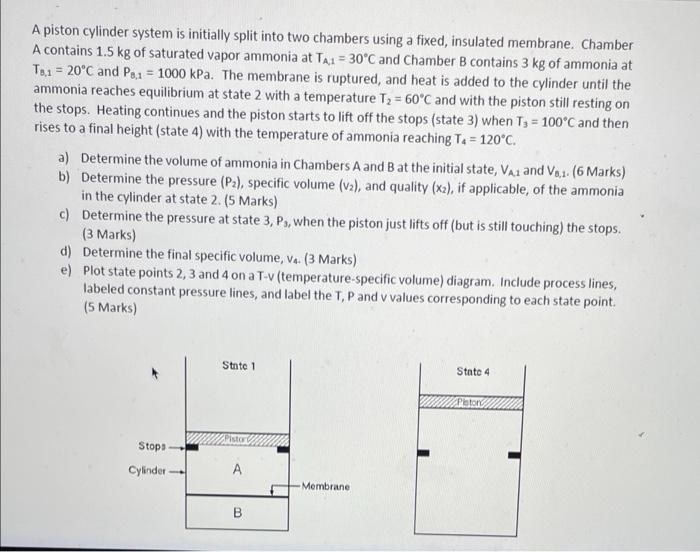
A piston cylinder system is initially split into two chambers using a fixed, insulated membrane. Chamber A contains 1.5 kg of saturated vapor ammonia at TA1 = 30C and Chamber B contains 3 kg of ammonia at T.1 = 20C and Po.1 = 1000 kPa. The membrane is ruptured, and heat is added to the cylinder until the ammonia reaches equilibrium at state 2 with a temperature T2 = 60'C and with the piston still resting on the stops. Heating continues and the piston starts to lift off the stops (state 3) when T3 = 100C and then rises to a final height (state 4) with the temperature of ammonia reaching Ta = 120'C. a) Determine the volume of ammonia in Chambers A and B at the initial state, Vaz and Vol. (6 Marks) b) Determine the pressure (P2), specific volume (v2), and quality (x2), if applicable, of the ammonia in the cylinder at state 2. (5 Marks) c) Determine the pressure at state 3, Ps, when the piston just lifts off (but is still touching) the stops. (3 Marks) d) Determine the final specific volume, Va. (3 Marks) e) Plot state points 2, 3 and 4 on a T-v (temperature-specific volume) diagram. Include process lines, labeled constant pressure lines, and label the T, P and v values corresponding to each state point (5 Marks) State 1 State 4 Pistor Stop Cylinder A A Membrane B 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


