Answered step by step
Verified Expert Solution
Question
1 Approved Answer
3. Ethanol is to be produced from ethylene and water using commercial catalyst in a packed bed flow reactor (PFR). The reaction is reversible as
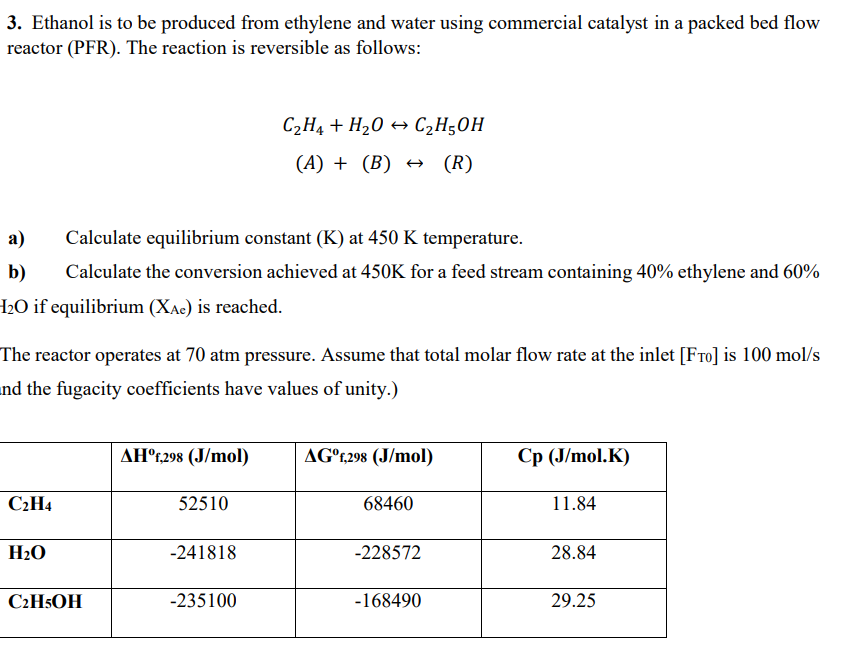 3. Ethanol is to be produced from ethylene and water using commercial catalyst in a packed bed flow reactor (PFR). The reaction is reversible as follows: C2H4+H2OC2H5OH(A)+(B)(R) a) Calculate equilibrium constant (K) at 450K temperature. b) Calculate the conversion achieved at 450K for a feed stream containing 40% ethylene and 60% I2O if equilibrium (XAc) is reached. The reactor operates at 70atm pressure. Assume that total molar flow rate at the inlet [FT0] is 100mol/s nd the fugacity coefficients have values of unity.)
3. Ethanol is to be produced from ethylene and water using commercial catalyst in a packed bed flow reactor (PFR). The reaction is reversible as follows: C2H4+H2OC2H5OH(A)+(B)(R) a) Calculate equilibrium constant (K) at 450K temperature. b) Calculate the conversion achieved at 450K for a feed stream containing 40% ethylene and 60% I2O if equilibrium (XAc) is reached. The reactor operates at 70atm pressure. Assume that total molar flow rate at the inlet [FT0] is 100mol/s nd the fugacity coefficients have values of unity.) Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


