Question
4. Correct acidic strength order of the given carbonyl compound is (I) (II) (III) Cl (IV) (a) IV > | > | > III
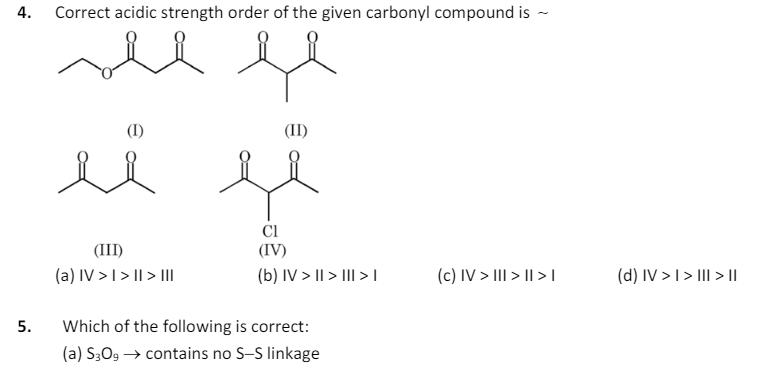

4. Correct acidic strength order of the given carbonyl compound is (I) (II) (III) Cl (IV) (a) IV > | > | > III (b) IV > II > III > 1 (c) IV > III > II >| (d) IV > | > III > II 5. Which of the following is correct: (a) S3O9 contains no S-S linkage -> (b) HNO4 contains no O-O linkage (c) (HPO3)3 contains P-P linkage (d) SO contains S-S linkage 6. At 373 K, a gaseous reaction A(g) 2B(g) + C(g) is found to be of first order. Starting with pure A, the total pressure at the end of 10 min was 176 mm Hg and after a long time when A was completely dissociated, it was 270 mm Hg. Then pressure of A at the end of 10 minutes was: (a) 94mm (b) 47mm (c) 43mm (d) 90mm
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access with AI-Powered Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


