Answered step by step
Verified Expert Solution
Question
1 Approved Answer
(5) A chemical analysis of water samnle hae log=0.5z21+activity=C=21zi2[C] (ii) What is the ionic strength of the solution? (a) 21,8 (b) 10.9 (c) 24.3 (d)
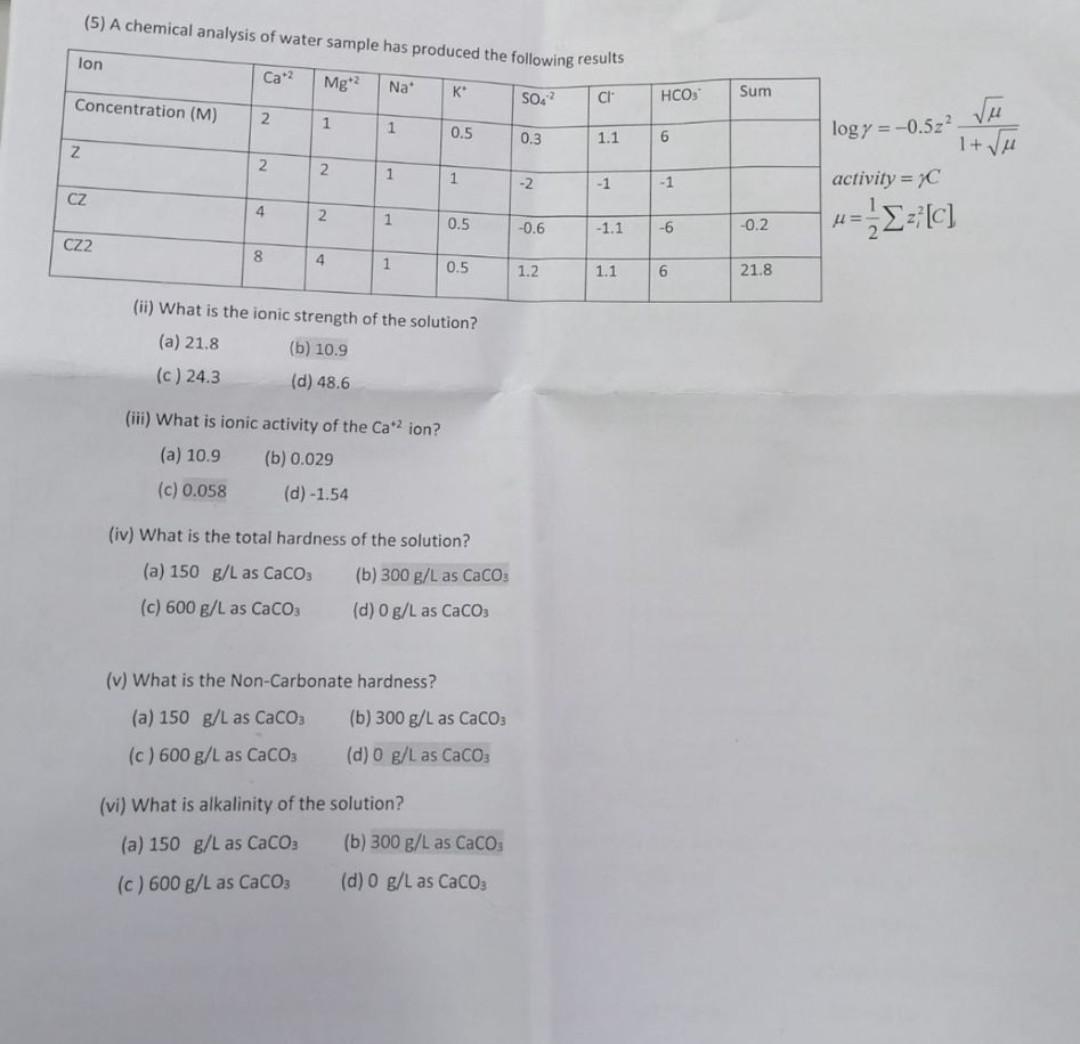
(5) A chemical analysis of water samnle hae log=0.5z21+activity=C=21zi2[C] (ii) What is the ionic strength of the solution? (a) 21,8 (b) 10.9 (c) 24.3 (d) 48.6 (iii) What is ionic activity of the Ca+2 ion? (a) 10.9 (b) 0.029 (c) 0.058 (d) -1.54 (iv) What is the total hardness of the solution? (a) 150g/L as CaCO3 (b) 300g/L as CaCO3 (c) 600g/L as CaCO3 (d) 0g/L as CaCO3 (v) What is the Non-Carbonate hardness? (a) 150g/L as CaCO3 (b) 300g/L as CaCO3 (c) 600g/L as CaCO3 (d) 0g/L as CaCO3 (vi) What is alkalinity of the solution? (a) 150g/L as CaCO3 (b) 300g/L as CaCO3 (c) 600g/L as CaCO3 (d) 0g/L as CaCO3
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


