Answered step by step
Verified Expert Solution
Question
1 Approved Answer
5. part A & B Ethyl chloride vapor decomposes by the fifst-order Find the value of the specific rate constant at 730K. reaction C2H5ClC2H4+HCl Express
5. part A & B 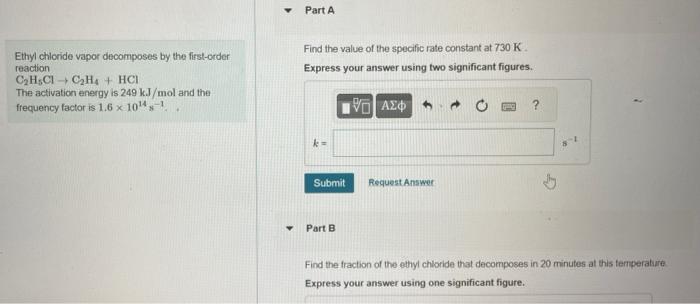
Ethyl chloride vapor decomposes by the fifst-order Find the value of the specific rate constant at 730K. reaction C2H5ClC2H4+HCl Express your answer using two significant figures. The activation energy is 249kJ/mol and the frequency factor is 1.61014s1. Part 8 Find the fraction of the ethyl chioride that decomposes in 20 minutes at this temperature Express your answer using one significant figure 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


