Question
** 6. HC-CH3 a. 7. 8. 9. 10. MeO a. a. Br a. @... OH Br CI 1. Br, hv 2. CHCC-, Et0 3.
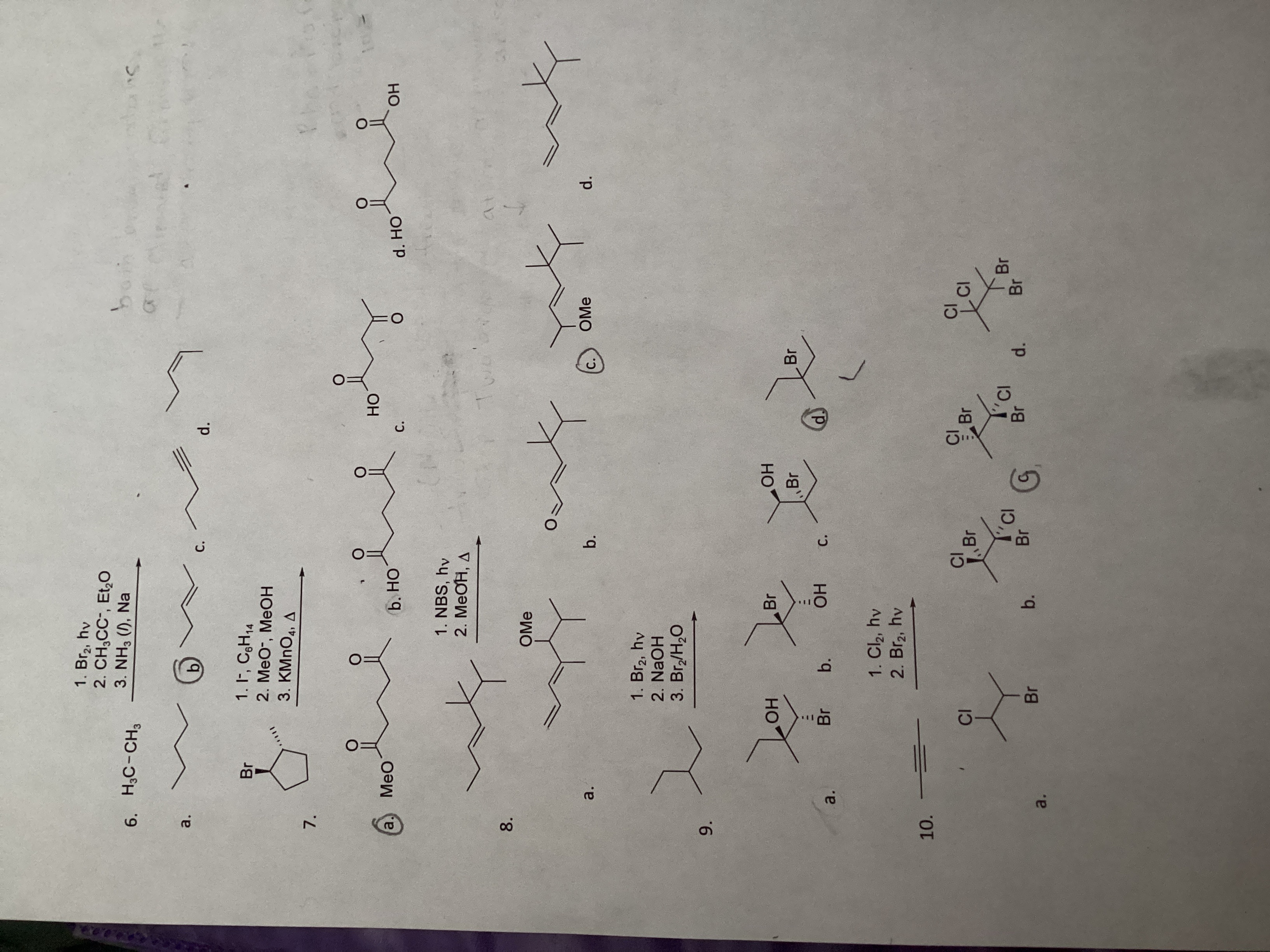
** 6. HC-CH3 a. 7. 8. 9. 10. MeO a. a. Br a. @... OH Br CI 1. Br, hv 2. CHCC-, Et0 3. NH3 (1), Na 1. I, C6H14 2. MeO-, MeOH 3. KMnO4, A b Br 1. Br, hv 2. NaOH 3. Br/HO b. b. HO 1. NBS, hv 2. MeOH, A OMe Br = OH 1. Cl, hv 2. Br, hv C. b. CI b. C. Br O Br CI OH Br G d. C. HO d Br Br CI C. Br d. OMe CI CI Br d. HO Br 1 d. OH S
Step by Step Solution
There are 3 Steps involved in it
Step: 1
Solutions g d Brg hr H BrBr Br Exor T...
Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get StartedRecommended Textbook for
Introductory Chemistry Concepts And Critical Thinking
Authors: Charles Corwin
7th Edition
0321804902, 9780321804907
Students also viewed these Chemical Engineering questions
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
View Answer in SolutionInn App



