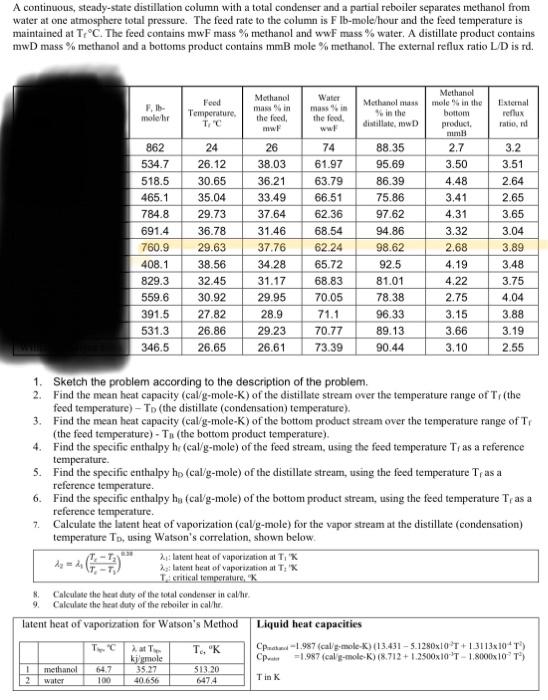
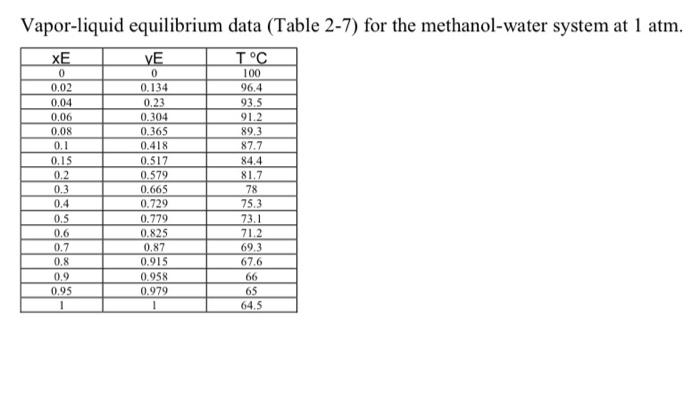
A continuous, steady-state distillation column with a total condenser and a partial reboiler separates methanol from water at one atmosphere total pressure. The feed rate to the column is F Ib-mole/hour and the feed temperature is maintained at TC. The feed contains mwF mass % methanol and wwf mass % water. A distillate product contains mwD mass % methanol and a bottoms product contains mmB mole % methanol. The external reflux ratio L/D is td. Food Temperature TC Methanol mawin the feed mw Water man in the food moler Methanol in the distillate, w Internal reflux ratio, 862 534.7 518.5 465.1 784.8 691.4 760.9 408.1 829.3 559.6 391.5 531.3 346.5 24 26.12 30.65 35.04 29.73 36.78 29.63 38.56 32.45 30.92 27.82 26.86 26.65 26 38.03 36.21 33.49 37.64 31.46 37.76 34.28 31.17 29.95 28.9 29.23 26.61 74 61.97 63.79 66.51 62.36 68.54 62.24 65.72 68.83 70.05 71.1 70.77 73.39 88.35 95.69 86.39 75.86 97.62 94.86 98.62 92.5 81.01 78.38 96.33 89.13 90.44 Methanol mole in the bottom product mumi 2.7 3.50 4.48 3.41 4.31 3.32 2.68 4.19 4.22 2.75 3.15 3.66 3.10 3.2 3.51 2.64 2.65 3.65 3.04 3.89 3.48 3.75 4.04 3.88 3.19 2.55 1. Sketch the problem according to the description of the problem, 2. Find the mean heat capacity (cal/g-mole-K) of the distillate stream over the temperature range of T (the feed temperature) - To the distillate (condensation) temperature). 3. Find the mean heat capacity (cal/g-mole-K) of the bottom product stream over the temperature range of T, (the feed temperature) - T (the bottom product temperature). 4. Find the specific enthalpy he (cal/g-mole) of the feed stream, using the feed temperature T, as a reference temperature 5. Find the specific enthalpy ho (cal/g-mole) of the distillate stream, using the feed temperature T, as a reference temperature 6. Find the specific enthalpy ha (cal/g-mole) of the bottom product stream, using the feed temperature Tras a reference temperature 7. Calculate the latent heat of vaporization (cal/g-mole) for the vapor stream at the distillate (condensation) temperature To, using Watson's correlation, shown below. 21:latent heat of vaporizational T, K == (1,7 Relatent het of vaporizational T, X TA critical temperature, Calculate the heat duty of the total condenser in calhe. 9. Calculate the heat duty of the reboiler in cal/l. latent heat of vaporization for Watson's Method Liquid heat capacities TC 2.at T. "K CP-1.987 (cal g-mole-K) 13.431 - 5.1280x1OT+13113x10T) ki gmole = 1.987 (cal g-mole-K) (8.712 +1.2500x10' 1.8000x10'') 35.27 -T d 1 2 methanol water 64.7 100 513.20 647.4 40.656 Tin K Vapor-liquid equilibrium data (Table 2-7) for the methanol-water system at 1 atm. XE VE TC O 0.02 0.04 0.06 0.08 0.1 0.15 0.2 0.3 0.4 0.5 0.6 0.7 0,8 0.9 0.95 1 0 0.134 0.23 0,304 0.365 0.418 0.517 0.579 0.665 0.729 0.779 0,825 0.87 0.915 0.958 0.979 1 100 96.4 93.5 91.2 89.3 87.7 84.4 81.7 78 75.3 73.1 71.2 69.3 67.6 66 65 64.5