Answered step by step
Verified Expert Solution
Question
1 Approved Answer
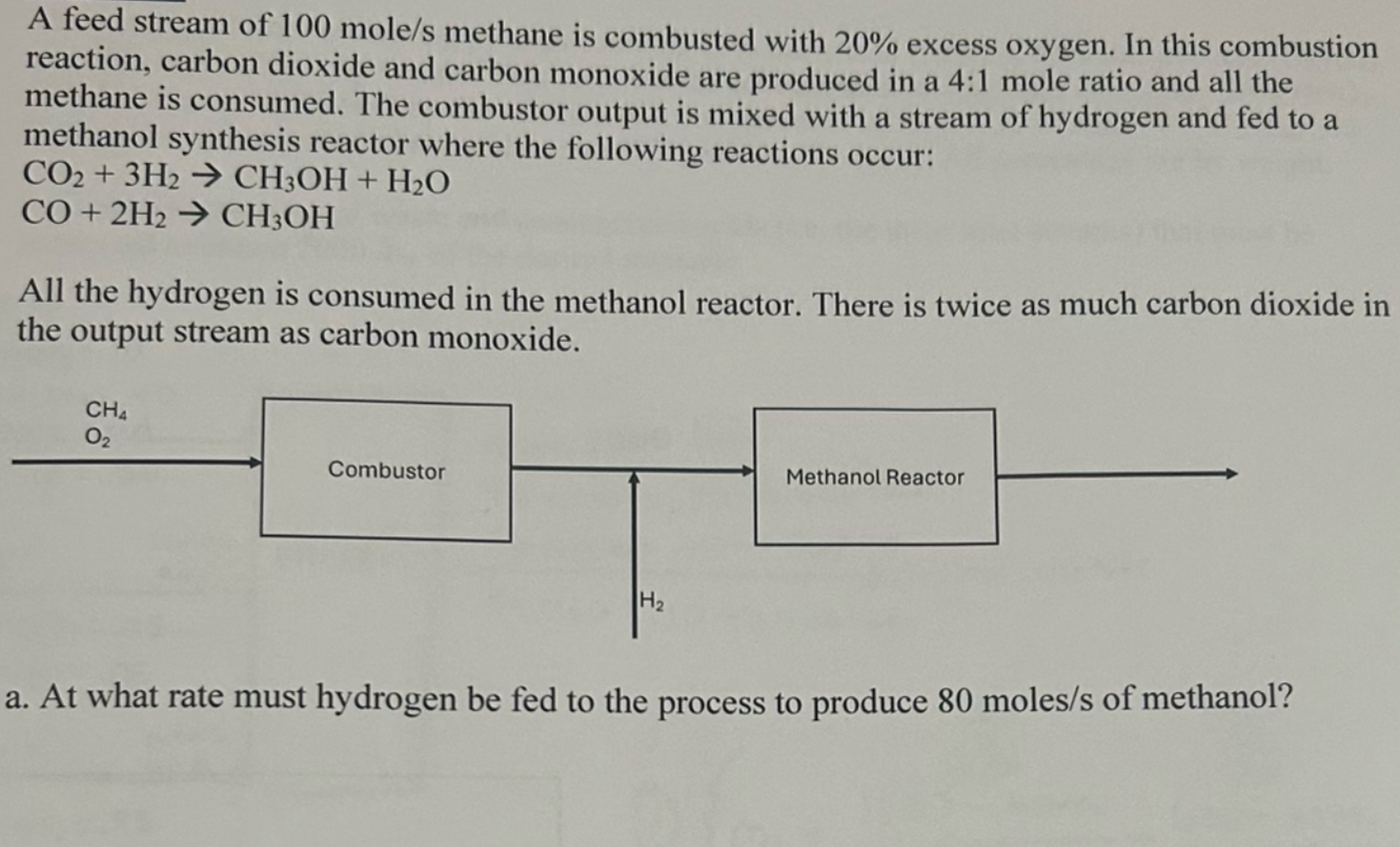
A feed stream of 1 0 0 mol / s methane is combusted with 2 0 % excess oxygen. In this combustion reaction, carbon dioxide
A feed stream of mols methane is combusted with excess oxygen. In this combustion reaction, carbon dioxide and carbon monoxide are produced in a : mole ratio and all the methane is consumed. The combustor output is mixed with a stream of hydrogen and fed to a methanol synthesis reactor where the following reactions occur:
All the hydrogen is consumed in the methanol reactor. There is twice as much carbon dioxide in the output stream as carbon monoxide.
a At what rate must hydrogen be fed to the process to produce molss of methanol?
b What is molar compositions YO YCO YCO, YHO and YCHOH of the output stream from methanol reactor?
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


