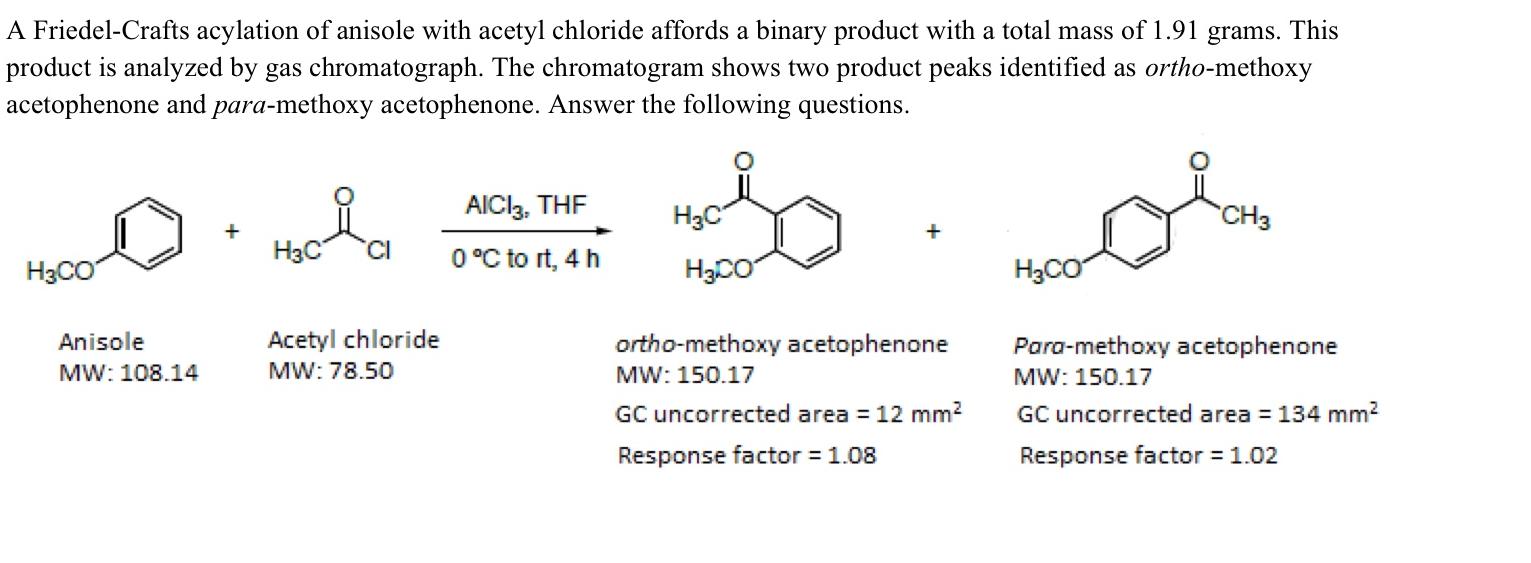
A Friedel-Crafts acylation of anisole with acetyl chloride affords a binary product with a total mass...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
A Friedel-Crafts acylation of anisole with acetyl chloride affords a binary product with a total mass of 1.91 grams. This product is analyzed by gas chromatograph. The chromatogram shows two product peaks identified as ortho-methoxy acetophenone and para-methoxy acetophenone. Answer the following questions. H3CO Anisole MW: 108.14 H3C Acetyl chloride MW: 78.50 AICI3, THF 0 °C to rt, 4 h H3C H3CO + ortho-methoxy acetophenone MW: 150.17 GC uncorrected area = 12 mm² Response factor = 1.08 H₂CO CH3 Para-methoxy acetophenone MW: 150.17 GC uncorrected area = 134 mm² Response factor = 1.02 A) Using all the data given above calculate the molar percent compositions of each of the two isomeric products obtained in the reaction. Also, calculate the mass of each of the two isomeric products in grams. Show all work. B) A student conducted the above reaction with 2.64 g of anisole and 2.36 g of acetyl chloride. He purified the product mixture by column chromatography and obtained 0.1254 g of ortho-methoxy acetophenone and 1.6843 g of para- methoxy acetophenone. Show how you would find out the limiting reagent. Show how you would calculate the number of moles of the materials needed. Calculate the theoretical yield and percent yield for each of the isomeric products. Show all calculations and write down all appropriate formulas. A Friedel-Crafts acylation of anisole with acetyl chloride affords a binary product with a total mass of 1.91 grams. This product is analyzed by gas chromatograph. The chromatogram shows two product peaks identified as ortho-methoxy acetophenone and para-methoxy acetophenone. Answer the following questions. H3CO Anisole MW: 108.14 H3C Acetyl chloride MW: 78.50 AICI3, THF 0 °C to rt, 4 h H3C H3CO + ortho-methoxy acetophenone MW: 150.17 GC uncorrected area = 12 mm² Response factor = 1.08 H₂CO CH3 Para-methoxy acetophenone MW: 150.17 GC uncorrected area = 134 mm² Response factor = 1.02 A) Using all the data given above calculate the molar percent compositions of each of the two isomeric products obtained in the reaction. Also, calculate the mass of each of the two isomeric products in grams. Show all work. B) A student conducted the above reaction with 2.64 g of anisole and 2.36 g of acetyl chloride. He purified the product mixture by column chromatography and obtained 0.1254 g of ortho-methoxy acetophenone and 1.6843 g of para- methoxy acetophenone. Show how you would find out the limiting reagent. Show how you would calculate the number of moles of the materials needed. Calculate the theoretical yield and percent yield for each of the isomeric products. Show all calculations and write down all appropriate formulas.
Expert Answer:
Answer rating: 100% (QA)
2 A Given total mass or the product area 12 mm Para pordnet area a 134 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Factor A Factor B 1 2 3 4 Xj for Factor B 1 2 3 4 1 2500 2 9 10 6 9 8500 3 14 11 16 12 13250 Xi for Factor A 8333 8000 9000 7333 X8083
-
The results taken from a mass spectrum of chlorine gas show peaks at m/z 35.00 and m/z 37.00 (The m/z peaks on a mass spectrum identify the different isotopes of an element that are present in the...
-
A rectangular aluminum bar 12 mm thick and 50 mm wide is welded to fixed supports at the ends, and the bar supports a load W = 3.5 kN, acting through a pin as shown. Find the reactions at the...
-
1-Calculate amount of secondary compression in terms of inches 10 years following end of the primary consolidation. S.sc ang (12)= Coefficient of secondary compression=0.035 Thickness of clay layer-...
-
Refer to the situation described in BE 6-6. How much will Leslie accumulate in three years by depositing $500 at the beginning of each of the next 12 quarters?
-
Southwest Transportation Inc. is considering a distribution facility at a cost of $10,000,000. The facility has an estimated life of 10 years and a $2,000,000 residual value. It is expected to...
-
Lance Lot and Arthur King are opening a FedEx Kinko's store. There are no competing copy shops in the area. Their fundamental decision is how to organize the business. Lot thinks the partnership form...
-
Thomson Company, a manufacturing firm, has supplied the following information from its accounting records for the last calendar year: Direct labor cost .................$371,500 Purchases of direct...
-
> The following figures represent the amount that can be produced with a fixed amount of factor inputs. Panama Honduras Bananas 100 160 Sugarcane 50 40 A. Which country has the absolute advantage in...
-
b. Prepare all consolidating entries needed to prepare consolidated statements for 20X5. (If no entry is required for a transaction/event, select "No journal entry required" in the first account...
-
The Engine Division of The Cleveland Automotive Corporation had sales of $7,200,000 and an operating income of $864,000 last year. The total assets of the Engine Division were $3,200,000 while...
-
Which sequences of paths are explored by BFS and DFS in this problem? Exercise 2 Given the graph represented in t Figure 1, where n, is the initial state, n, is the final state, each edge nn' is...
-
2. The spring has a spring constant of k = 1.4 kN/m and an unstretched length of r = 400 mm. Determine the force in the spring when theta is 55 degrees. Also determine the scaler x and y components...
-
Would the outcome be the same or different in Turkey than in the Article Is Monitoring Employee Communications Lawful? Explain in detail.
-
Mike Ibanez had a big decision to make. He had inherited his family's vegetable business. Ibanez Produce grew, harvested, packaged, and sold produce. Mike was concerned because a large storm was...
-
Describe a federal law that shapes discretionary benefits practices and identify the relevant employee benefits practices to which they apply. What are pros and cons of this law?
-
Examine the image representing a straddle position. The image represents the value of 10 June call options and 10 June put options all with a strike price of $75. The investor paid $9590 to establish...
-
Critical reading SAT scores are distributed as N(500, 100). a. Find the SAT score at the 75th percentile. b. Find the SAT score at the 25th percentile. c. Find the interquartile range for SAT scores....
-
As a method for the preparation of alkenes, a weakness in the acid-catalyzed dehydration of alcohols is that the initially formed alkene (or mixture of alkenes) sometimes isomerizes under the...
-
Select the combination of alkyl bromide and potassium alkoxide that would be the most effective in the syntheses of the following ethers: (a) CH3OC(CH3)3 (b) (c) (CH3)3CCH2OCH2CH3 OCH
-
Write chemical equations for the reaction that takes place between each of the following pairs of reactants: (a) 3-Ethyl-3-pentanol and hydrogen chloride (b) 1-Tetradecanol and hydrogen bromide
-
Describe the evolution of specifications from initial System Concept to SPS.
-
What is a Specification Tree and how is it structured?
-
How does each type of specification apply to system development?

Study smarter with the SolutionInn App


