Answered step by step
Verified Expert Solution
Question
1 Approved Answer
A liquid containing 25 mol% toluene, 40 mol% ethylbenzene, and the balance water, is subjected to flash distillation at a total pressure of 0.5 atm.
A liquid containing 25 mol% toluene, 40 mol% ethylbenzene, and the balance water, is subjected to flash distillation at a total pressure of 0.5 atm. The vapor pressure data of the substances are found in the table below. Assuming that the ethylbenzene-toluene mixture obeys Raoult's law and that the hydrocarbons are completely immiscible in water, calculate the temperature and composition of the liquid and vapor phases at a) their bubble point, b) their dew point, c) 50% vaporized fraction. Use excel
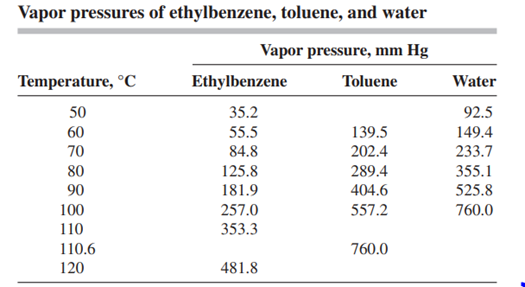
Vapor pressures of ethylbenzene, toluene, and water Water Temperature, C 50 60 70 80 90 100 110 110.6 120 Vapor pressure, mm Hg Ethylbenzene Toluene 35.2 55.5 139.5 84.8 202.4 125.8 289.4 181.9 404.6 257.0 557.2 353.3 760.0 481.8 92.5 149.4 233.7 355.1 525.8 760.0
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


