Question
A nickel-manganese battery is constructed by immersing bars of nickel and manganese in slightly acidic (pH ~ 3) solutions of their own ions, separated by
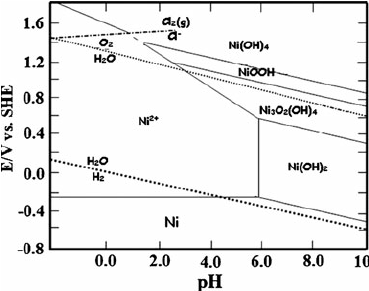 A nickel-manganese battery is constructed by immersing bars of nickel and manganese in slightly acidic (pH ~ 3) solutions of their own ions, separated by a porous membrane so that no interdiffusion of metallic ions occurs.
A nickel-manganese battery is constructed by immersing bars of nickel and manganese in slightly acidic (pH ~ 3) solutions of their own ions, separated by a porous membrane so that no interdiffusion of metallic ions occurs.
a. Draw a schematic of this battery. Use the attached Pourbaix diagram and discuss what reactions are likely to occur at the electrodes. Determine in what direction these reactions would proceed during discharge of the battery. Calculate the initial voltage (EMF) generated by the battery.
b. The battery drives a load that absorbs 2.5 A. Sketch a qualitative Evans E log j diagram that describes this process and calculate the equivalent resistance of the load.
c. Calculate the decrease in the EMF after one hour of operation at constant current. Explain why the current cannot be kept constant for a fixed load.
d. Describe how the battery can be recharged.
Data: Metal ion concentrations in the respective solutions: [Ni2+] = 0.5 M, [Mn2+] = 10-3 M. Areas of the electrodes: Ni: 100 cm2 , Mn: 50 cm2 Tafel slopes d/d(logj) (in Volt/decade) Ni2+/Ni: 0.12, Mn2+/Mn: 0.12 Exchange current densities (in A/cm2 ) Ni2+/Ni: 10-5 , Mn2+/Mn: 10-3
Ni(OH) NICOH NisOz(OH) E/V vs. SHE Ni(OH) VB 0.0 2.0 4.0 pH 6.0 8.0 10 Ni(OH) NICOH NisOz(OH) E/V vs. SHE Ni(OH) VB 0.0 2.0 4.0 pH 6.0 8.0 10Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


