Answered step by step
Verified Expert Solution
Question
1 Approved Answer
A pure substance X flows through a heat exchanger under steady conditions as shown in the figure below. The inlet temperature and pressure are 350K
A pure substance X flows through a heat exchanger under steady conditions as shown in the figure below. The inlet temperature and pressure are 350K and 8 bars, respectively, and outlet temperature and pressure are 250 K and 1 bar, respectively.
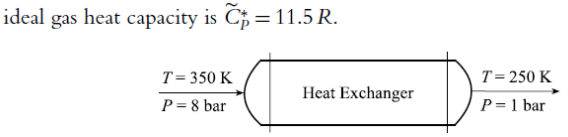
Estimate the amount of heat added to or removed from substance X using the following information provided from the solutions of the PengRobinson equation of state at the inlet and outlet conditions:
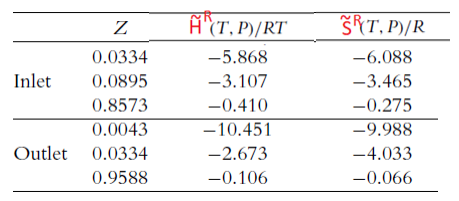
MAKE SURE TO GET AN ANSWER OF Q = -30000 J/mol!!!!
CP=11.5R \begin{tabular}{lccc} \hline & Z & H~R(T,P)/RT & S~R(T,P)/R \\ \hline \multirow{3}{*}{ Inlet } & 0.0334 & 5.868 & 6.088 \\ & 0.0895 & 3.107 & 3.465 \\ & 0.8573 & 0.410 & 0.275 \\ \hline \multirow{3}{*}{ Outlet } & 0.0043 & 10.451 & 9.988 \\ & 0.0334 & 2.673 & 4.033 \\ & 0.9588 & 0.106 & 0.066 \\ \hline \end{tabular}Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


