Answered step by step
Verified Expert Solution
Question
1 Approved Answer
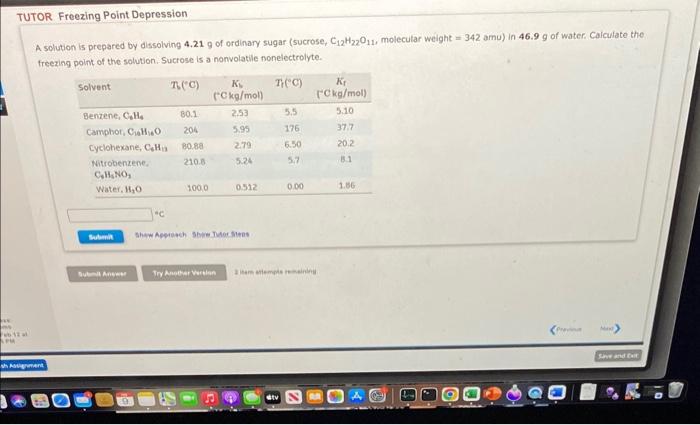
A solution is prepared by dissolving 4.21g of ordinary sugar (sucrose, C12H22O11, molecular weight =342amu ) in 46.9g of water. Calculate the. freezing point of
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


