Answered step by step
Verified Expert Solution
Question
1 Approved Answer
A,B,C,D In Section 11.5 in the textbook we defined the vapor pressure of a liquid in terms of an equilibrium. Write the equation representing the
A,B,C,D 
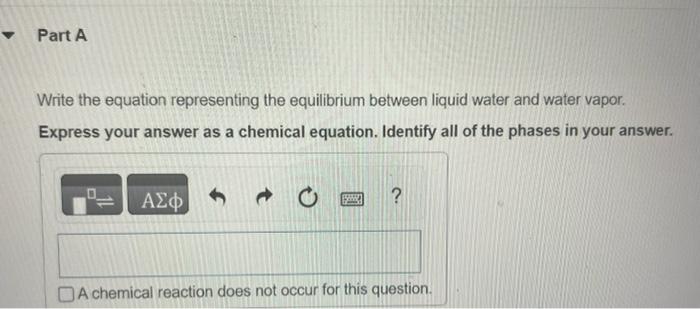
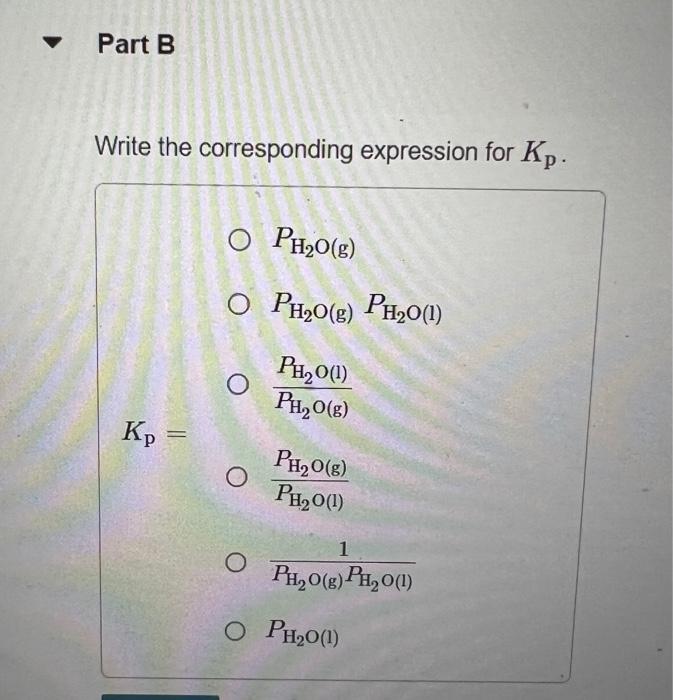
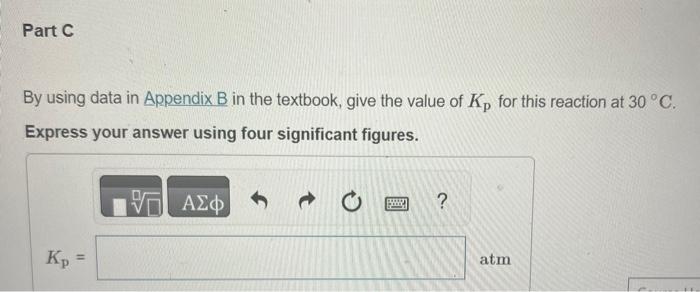
 In Section 11.5 in the textbook we defined the vapor pressure of a liquid in terms of an equilibrium. Write the equation representing the equilibrium between liquid water and water vapor. Express your answer as a chemical equation. Identify all of the phases in your answer. Write the corresponding expression for Kp. PH2O(g)PH2O(g)PH2O(l)PH2O(g)PH2O(l)Kp=PH2O(l)PH2O(g)PH2O(g)PH2O(l)1PH2O(l) By using data in Appendix B in the textbook, give the value of Kp for this reaction at 30C. Express your answer using four significant figures. What is the value of Kp for any liquid in equilibrium with its vapor at the normal boiling point of the liquid? Express your answer using three significant figures
In Section 11.5 in the textbook we defined the vapor pressure of a liquid in terms of an equilibrium. Write the equation representing the equilibrium between liquid water and water vapor. Express your answer as a chemical equation. Identify all of the phases in your answer. Write the corresponding expression for Kp. PH2O(g)PH2O(g)PH2O(l)PH2O(g)PH2O(l)Kp=PH2O(l)PH2O(g)PH2O(g)PH2O(l)1PH2O(l) By using data in Appendix B in the textbook, give the value of Kp for this reaction at 30C. Express your answer using four significant figures. What is the value of Kp for any liquid in equilibrium with its vapor at the normal boiling point of the liquid? Express your answer using three significant figures





Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


