Answered step by step
Verified Expert Solution
Question
1 Approved Answer
ANSWER BOTH OF THE FOLLOWING QUESTIONS Q.11 : For each of the following particles, do the following: a. BCl3 b.BH4c.H2S d. CF4 b) Name the
ANSWER BOTH OF THE FOLLOWING QUESTIONS 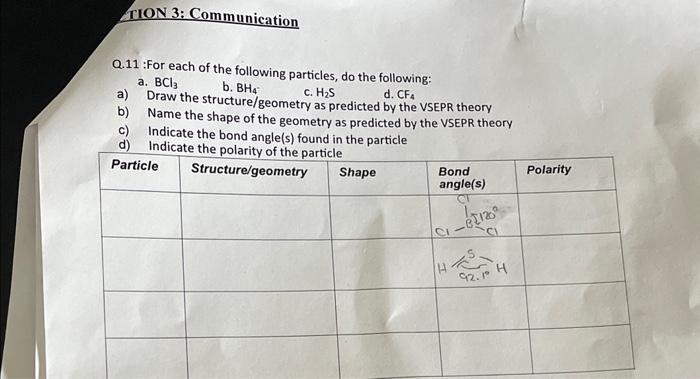

Q.11 : For each of the following particles, do the following: a. BCl3 b.BH4c.H2S d. CF4 b) Name the structure/geometry as predicted by the VSEPR theory b) Name the shape of the geometry as predicted by the VSEPR theory c) Indicate the bond angle(s) found in the particle Q.9-Draw a Bohr-rutherford diagram of an excited fluorine atom. Also explain ground state 

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


