Answered step by step
Verified Expert Solution
Question
1 Approved Answer
answer both questions A reaction was shown to follow second-order kinetics. How much time is required for [A] to change from 0.500M to 0.270M ?
answer both questions 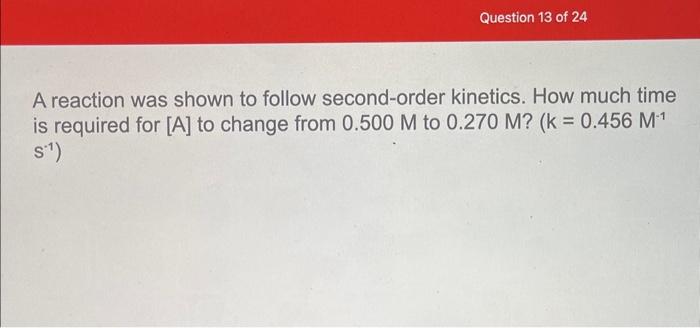

A reaction was shown to follow second-order kinetics. How much time is required for [A] to change from 0.500M to 0.270M ? (k=0.456M1 s1 ) A reaction has a rate law of Rate =(1.25M1s1)[A][B]. What is the rate of the reaction if [A]=0.591M and [B]=0.280M 

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


