Answered step by step
Verified Expert Solution
Question
1 Approved Answer
At room temperature the irreversible second order liquid phase reaction proceeds as follows: A batch reactor takes 18 minutes to fill and empty. What percentage
At room temperature the irreversible second order liquid phase reaction proceeds as follows:
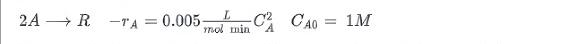 A batch reactor takes 18 minutes to fill and empty. What percentage conversion and reaction time should be used to maximize the daily product output R?
A batch reactor takes 18 minutes to fill and empty. What percentage conversion and reaction time should be used to maximize the daily product output R?
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


