Answered step by step
Verified Expert Solution
Question
1 Approved Answer
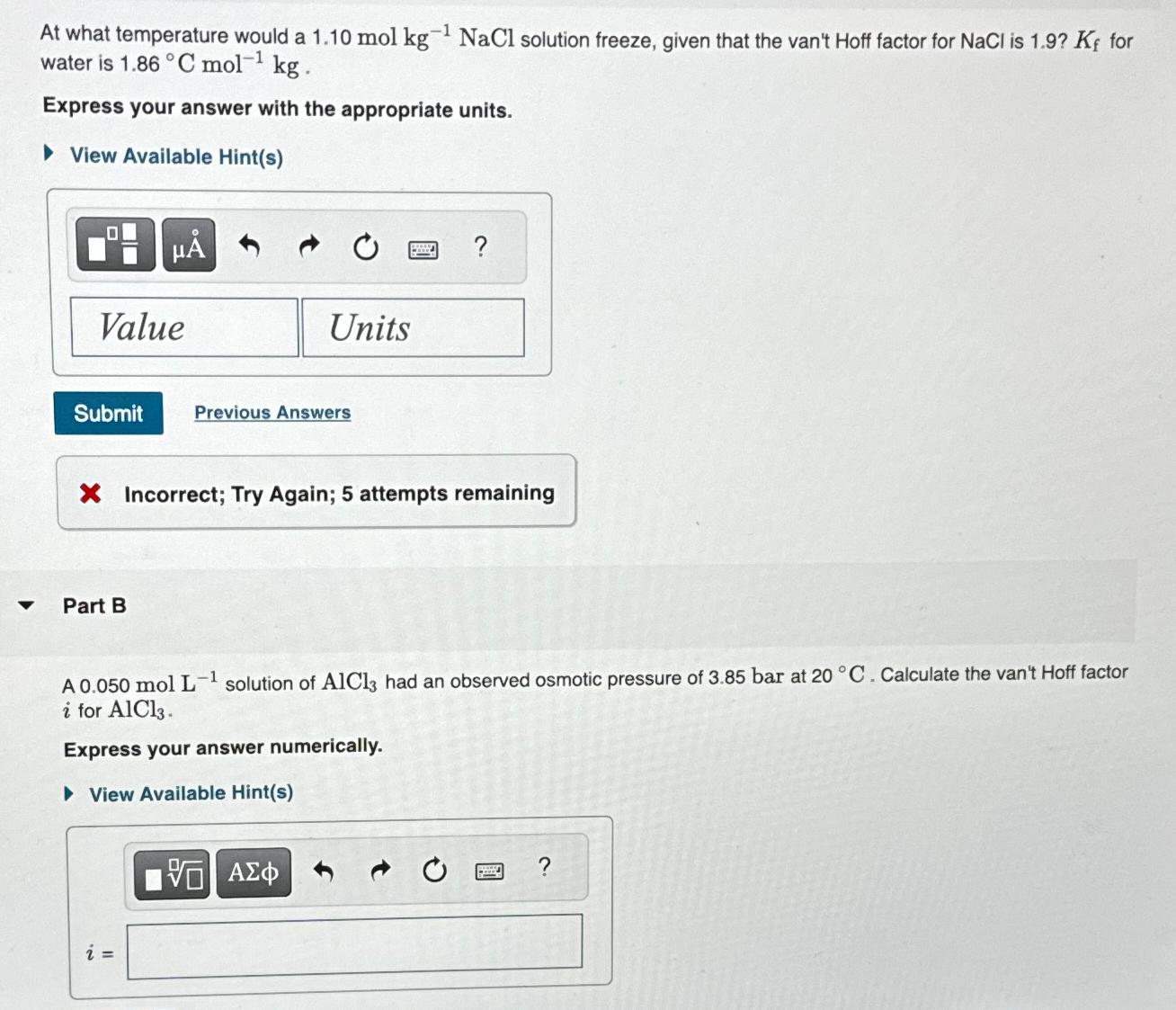
At what temperature would a 1 . 1 0 m o l k g k g - 1 NaCl solution freeze, given that the van't
At what temperature would a NaCl solution freeze, given that the van't Hoff factor for NaCl is for water is
Express your answer with the appropriate units.
View Available Hints
Previous Answers
Incorrect; Try Again; attempts remaining
Part B
A molL solution of had an observed osmotic pressure of bar at Calculate the van't Hoff factor i for
Express your answer numerically.
View Available Hints
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


