Answered step by step
Verified Expert Solution
Question
1 Approved Answer
b The reaction 2NO(g)+O2(g)+2NO2(g) is second order in NO and first conder in O2. When [NO]=0.4M and [O2]2.8M, the observed rate of the reaction is
b 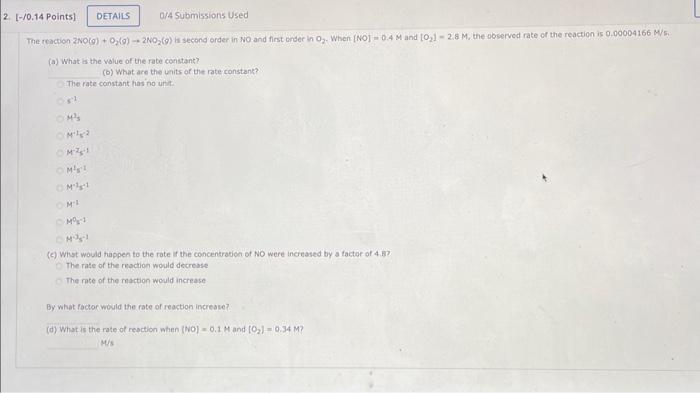
The reaction 2NO(g)+O2(g)+2NO2(g) is second order in NO and first conder in O2. When [NO]=0.4M and [O2]2.8M, the observed rate of the reaction is 0.00004166M/s. (a) What is the volue of the rate constant? (b) What are the units of the rate constank? The rate constant has ino unit. s1 M1s M1s2 M2s1 Ms1 M1s1 M1 M0s1 M1s1 (c). What would happen to the rote ir the concentraticn of No were increased by a factor of 4. as? The rate of the reaction would decresse The rate of the reartion would increase By what roctor would the rate of reaction increase? (d) What is the rate of repction when (NO)=0.1M and [O2]=0.34M ? M/s 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


