Question
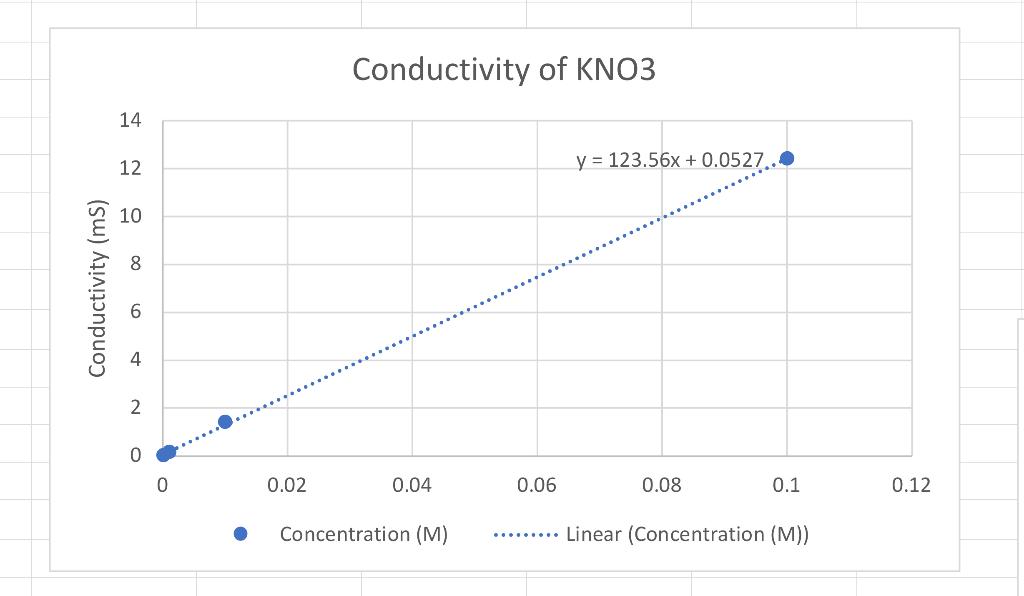
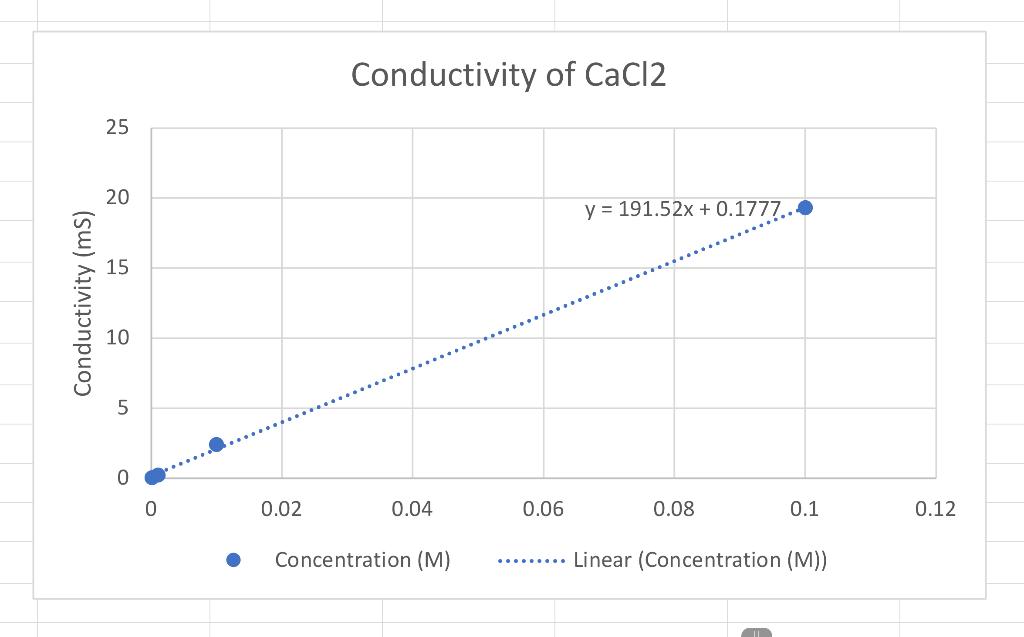
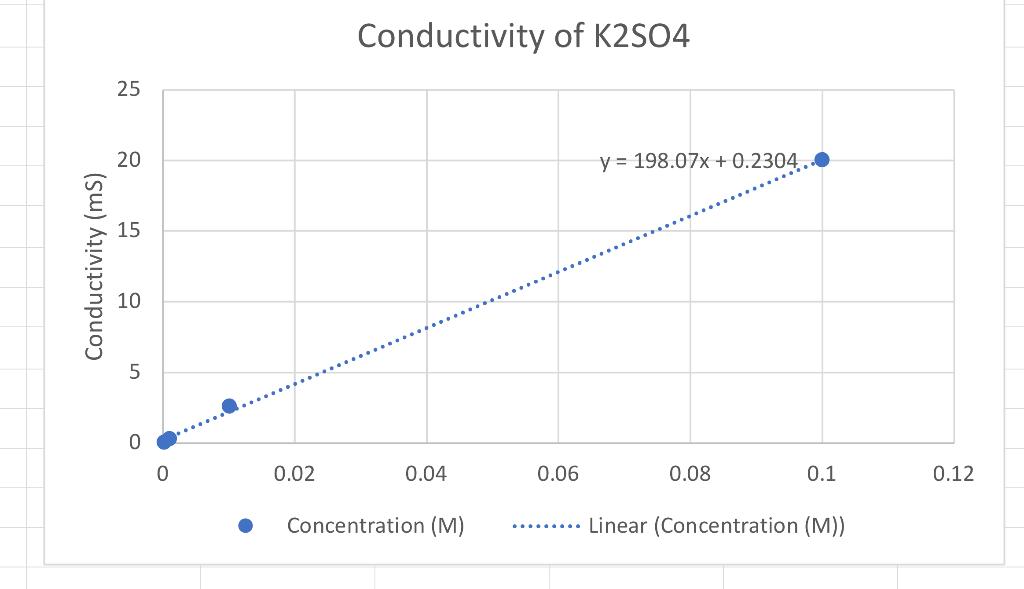
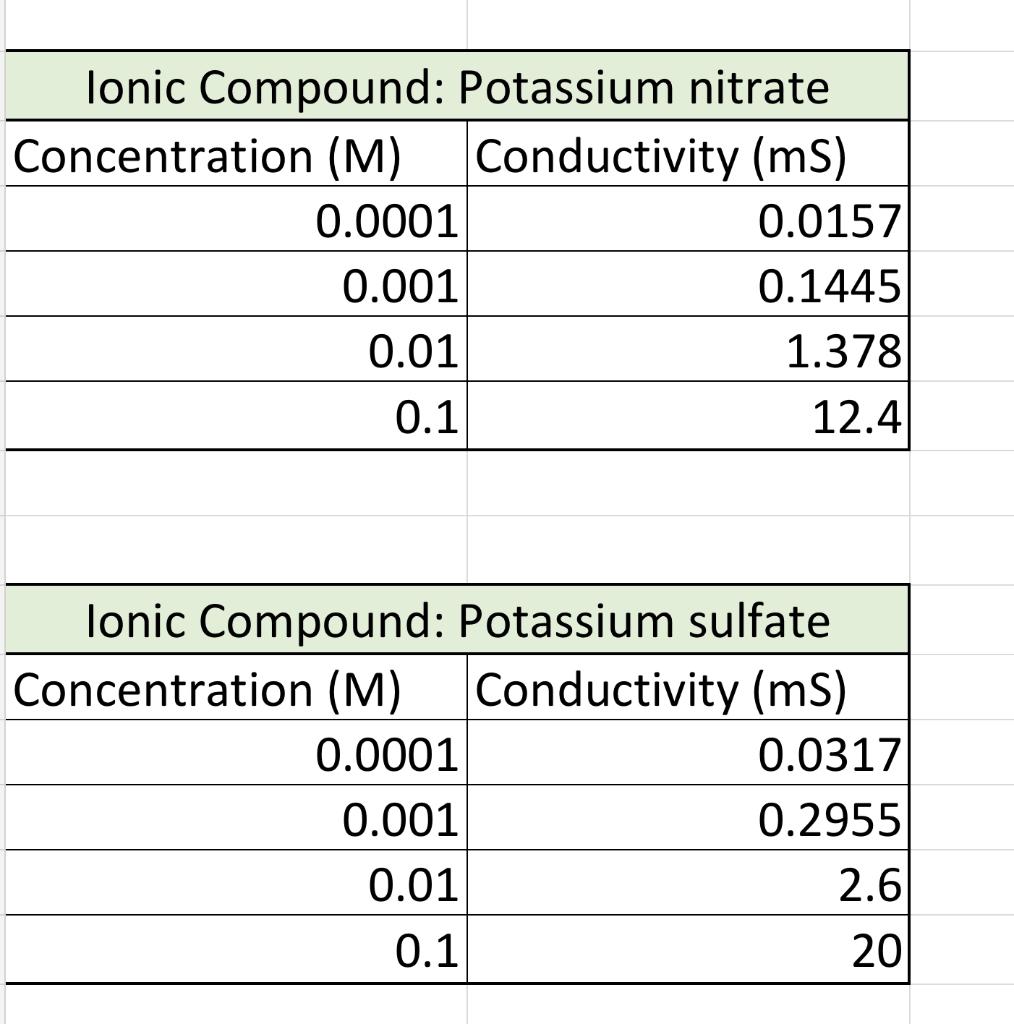
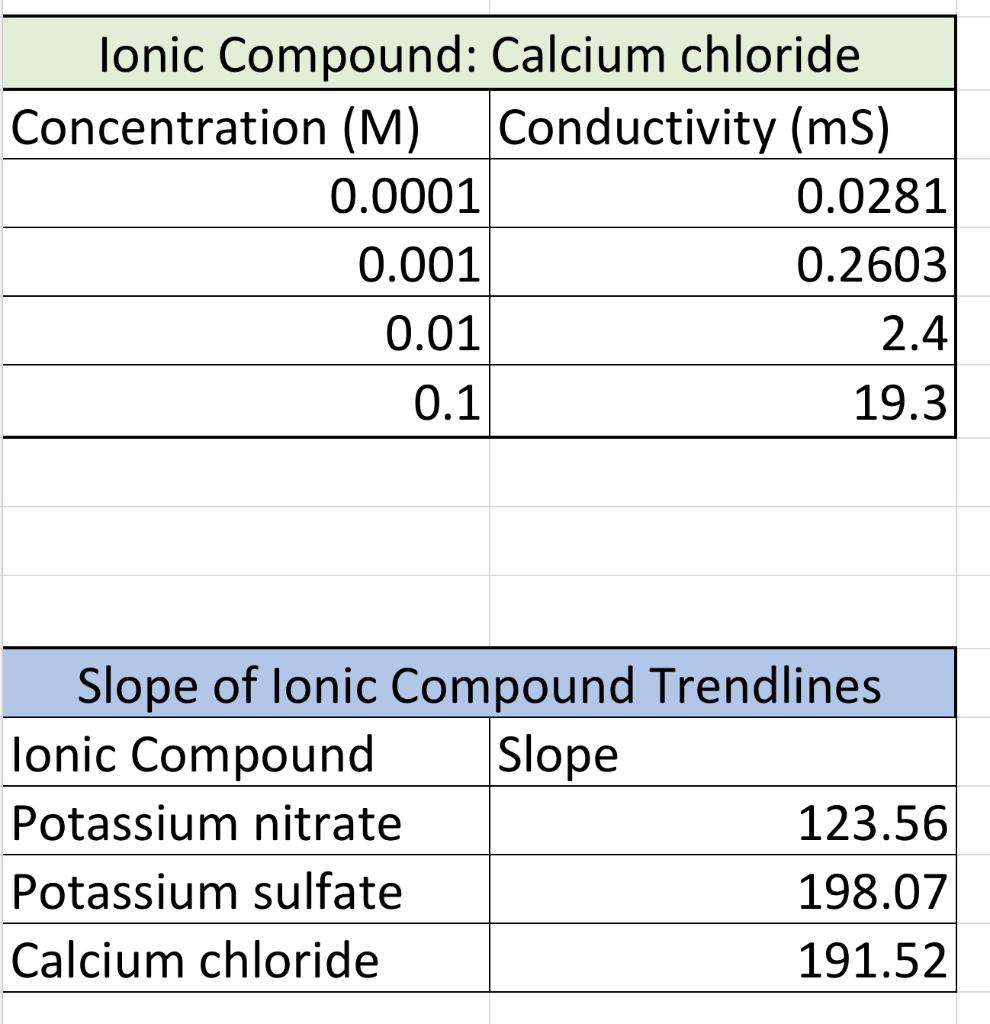
Based on the conductivity lab results answer the following questions please. 1.Which graph had the steepest slope? The smallest? 2.Given the equation for the dissociation
Based on the conductivity lab results answer the following questions please.
1.Which graph had the steepest slope? The smallest?
2.Given the equation for the dissociation of Li, CO, below, write the equation for the dissociation of the ionic compounds in this lab. (Don't worry about subscripts & superscripts) Li, CO, > 2Li+Co,2 11 12 Answer: a) KNO, -> b) K2S02-> c)CaCi2->
3. Since all solutions had the same concentration, what accounts for the difference in the slope of the three plots? Explain well. Answer:
4. If you repeated this experiment with MgS04, would you expect the slope of the line to be close to that of potassium nitrate or potassium sulfate? Explain well. Answer:
5. In another conductiviy lab of solution, NaOH conducts electricity but sugar does not. Explain well. (Hint: what's different about these two molecules chemically?)
Answer:
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


