Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Can please solve it for me? thank you At 40C,H2O2(aq) will decompose according to the following reaction: 2H2O2(aq)2H2O(l)+O2(g) The following data were collected for the
Can please solve it for me? thank you 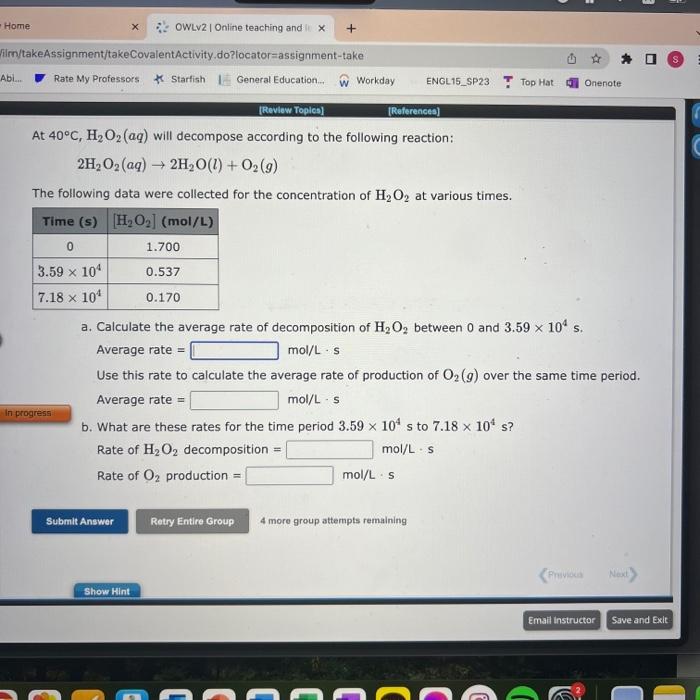
At 40C,H2O2(aq) will decompose according to the following reaction: 2H2O2(aq)2H2O(l)+O2(g) The following data were collected for the concentration of H2O2 at various times. a. Calculate the average rate of decomposition of H2O2 between 0 and 3.59104s. Average rate =mol/Ls Use this rate to calculate the average rate of production of O2(g) over the same time period. Average rate =mol/Ls b. What are these rates for the time period 3.59104s to 7.18104s ? Rate of H2O2 decomposition = mol/Ls Rate of O2 production =mol/Ls 4 more group attempts remaining 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


