Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Can someone help me to answer this correctly, please, and explain it. Please answer these 3 short questions, I don't have more money to buy
Can someone help me to answer this correctly, please, and explain it. Please answer these 3 short questions, I don't have more money to buy more questions. These 3 questions are different.
1. a.

1. b.

1. c.
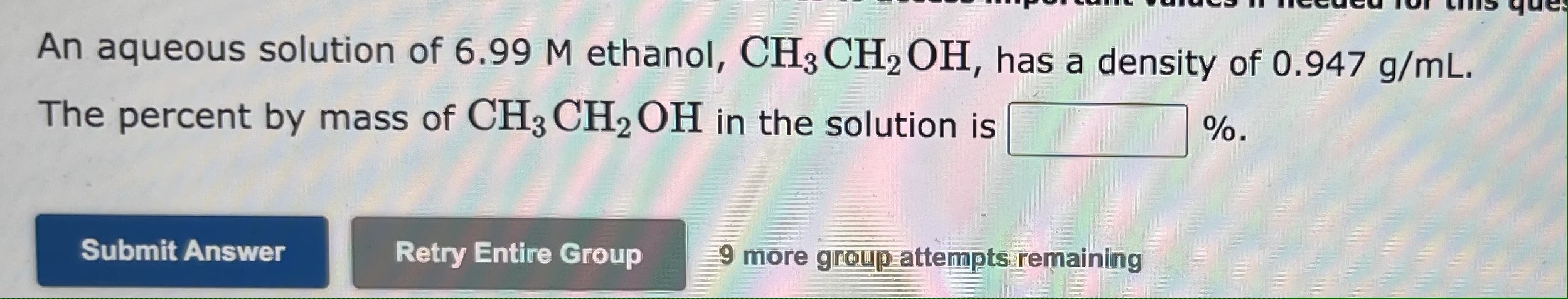
An aqueous solution is 34.0% by mass ethanol, CH3CH2OH, and has a density of 0.947g/mL. The mole fraction of ethanol in the solution is The mole fraction of calcium chloride, CaCl2, in an aqueous solution is 0.0271. The percent by mass of calcium chloride in the solution is % An aqueous solution of 6.99M ethanol, CH3CH2OH, has a density of 0.947g/mL. The percent by mass of CH3CH2OH in the solution is %. 9 more group attempts remaining
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


