Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Cd | CdCl2| Hg,Cl2|Hg (Cd+Hg,C1(s)=Cd +221 +2Hg(1)). E==0.671.02X 10 (T298)2.4x10 (T-298). Calculate 4,69, A,Hand A, So when the temperature is 313 K
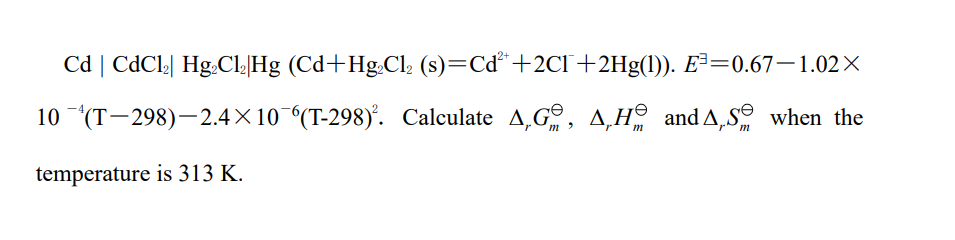
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


