Answered step by step
Verified Expert Solution
Question
1 Approved Answer
CH.5- 14, 18, 19 could you please help me get the correct answer i only have one chance left 14 10 points Enter your answer
CH.5- 14, 18, 19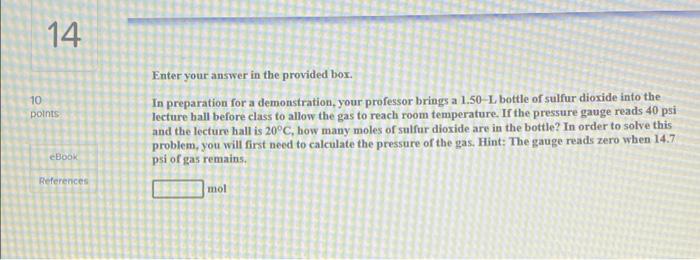


14 10 points Enter your answer in the provided box. In preparation for a demonstration, your professor brings a 1.50-L bottle of sulfur dioxide into the lecture ball before class to allow the gas to reach room temperature. If the pressure gauge reads 40 psi and the lecture ball is 20C, how many moles of sulfur dioxide are in the bottle? In order to solve this problem, you will first need to calculate the pressure of the gas, Hint: The gauge reads zero when 14.7 psi of gas remains. eBook References mol 18 10 points Enter your answer in the provided box. After 0.600 L of Ar at 1.36 atm and 233C is mixed with 0.200 L of O, at 504 torr and 144C in a 400-ml flask at 28C, what is the pressure in the lask? eBook atu References 19 10 Doints Enter your answer in the provided box. How many grams of potassium chlorate decompose to potassium chloride and 421 mL of O, at 128C and 738 torr? 2 KC103(3) - 2 KCK(s) + 3 02 (8) eBook References could you please help me get the correct answer i only have one chance left



Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


