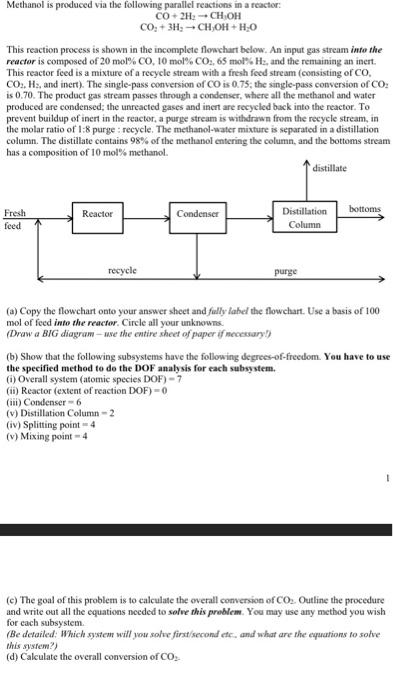
CO+2H2CH1OHCO2+3H2CH3OH+H2O This reaction process is shown in the incomplete flowchart below. An input gas stream into the reactor is composed of 20mol%CO210 mol\% CO2,65 mol\% H, and the remaining an inert. This reactor feed is a mixture of a recycle stream with a fresh feed stream (consisting of CO, CO2,H2, and inert). The single-pass conversion of CO is 0.75; the single-pass conversion of CO2 is 0.70. The product gas stream passes through a condenser, where all the methanol and water produced are condensed; the unreacted gases and inent are recyeled back into the reactor. To prevent buildup of inert in the reactor, a parge stream is withdrawn from the recycle stream, in the molar ratio of 1:8 purge ; recycle. The methanod-water mixture is separated in a distillation column. The distillate contains 98% of the methanol entering the column, and the bottoms stream has a composition of 10mol% methanol. (a) Copy the flowchart onte your answer sheet and fally lahel the flowehart. Use a basis of 100 mol of feed into the reactor. Circle all your unknowns. (Draw a BIG diagram - use the entire sheet of paper if mecesary?) (b) Show that the following subsystems have the following degrees-of-freedom. You have to use the specified methed to do the DOF analysis for each subsystem. (i) Overall system (atomic species DOF) =7 (ii) Reactor (extent of reaction DOF) =0 (iii) Condenser =6 (v) Distillation Column =2 (iv) Splitting point =4 (v) Mixing point =4 (c) The goal of this problem is to calculate the overall conversion of CO2. Outline the procedure and write out all the equations needed to solve this problem. You may use any method you wish for each subsystem. (Be detailed: Which system will you solve first/second etes and what are the equations to sohve this sysrem?) (d) Calculate the overall conversion of CO2 - CO+2H2CH1OHCO2+3H2CH3OH+H2O This reaction process is shown in the incomplete flowchart below. An input gas stream into the reactor is composed of 20mol%CO210 mol\% CO2,65 mol\% H, and the remaining an inert. This reactor feed is a mixture of a recycle stream with a fresh feed stream (consisting of CO, CO2,H2, and inert). The single-pass conversion of CO is 0.75; the single-pass conversion of CO2 is 0.70. The product gas stream passes through a condenser, where all the methanol and water produced are condensed; the unreacted gases and inent are recyeled back into the reactor. To prevent buildup of inert in the reactor, a parge stream is withdrawn from the recycle stream, in the molar ratio of 1:8 purge ; recycle. The methanod-water mixture is separated in a distillation column. The distillate contains 98% of the methanol entering the column, and the bottoms stream has a composition of 10mol% methanol. (a) Copy the flowchart onte your answer sheet and fally lahel the flowehart. Use a basis of 100 mol of feed into the reactor. Circle all your unknowns. (Draw a BIG diagram - use the entire sheet of paper if mecesary?) (b) Show that the following subsystems have the following degrees-of-freedom. You have to use the specified methed to do the DOF analysis for each subsystem. (i) Overall system (atomic species DOF) =7 (ii) Reactor (extent of reaction DOF) =0 (iii) Condenser =6 (v) Distillation Column =2 (iv) Splitting point =4 (v) Mixing point =4 (c) The goal of this problem is to calculate the overall conversion of CO2. Outline the procedure and write out all the equations needed to solve this problem. You may use any method you wish for each subsystem. (Be detailed: Which system will you solve first/second etes and what are the equations to sohve this sysrem?) (d) Calculate the overall conversion of CO2