An ideal gas (the weight is m kg) in an idealized piston-cylinder assembly undergoes a series...
Fantastic news! We've Found the answer you've been seeking!
Question:
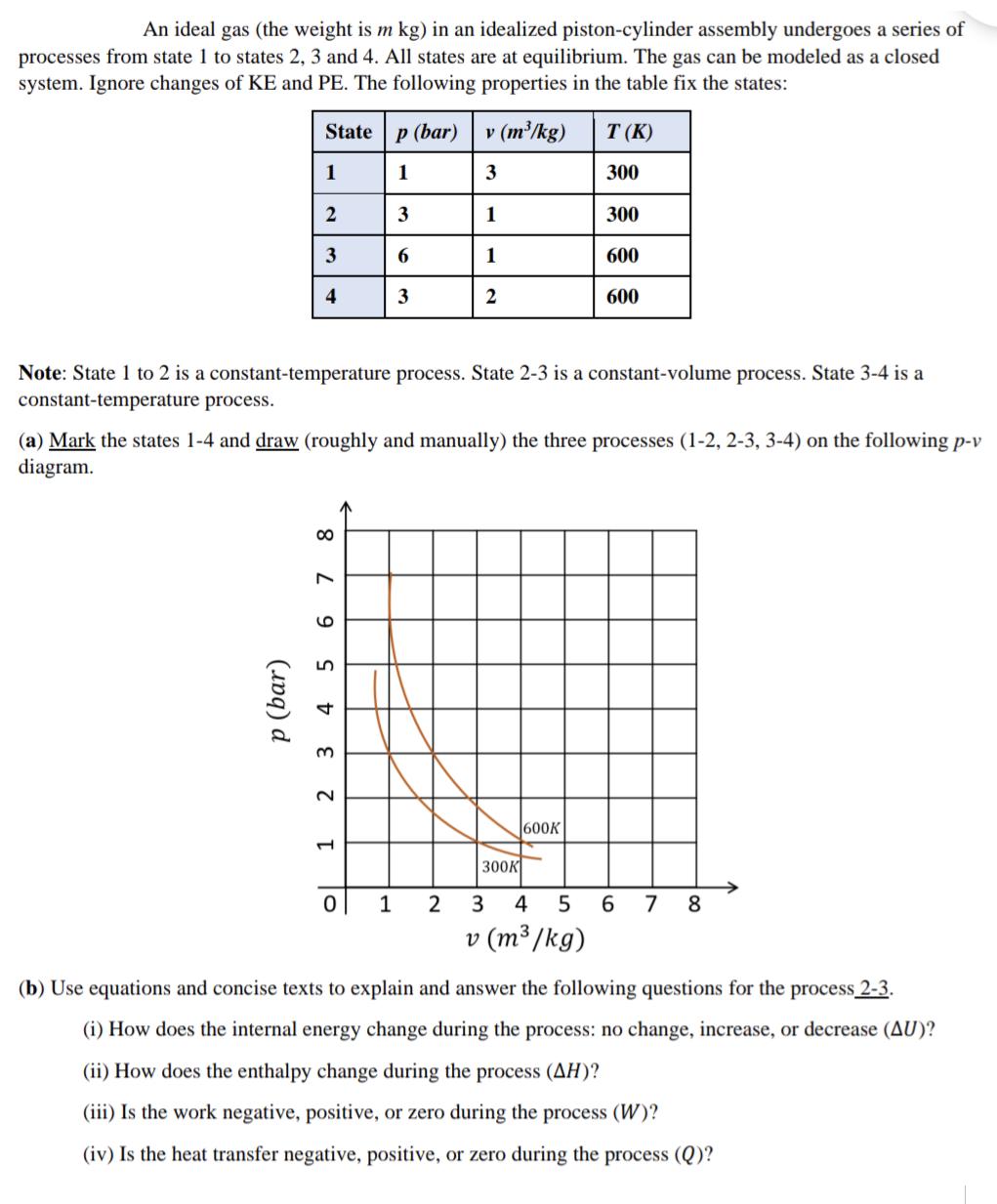
Transcribed Image Text:
An ideal gas (the weight is m kg) in an idealized piston-cylinder assembly undergoes a series of processes from state 1 to states 2, 3 and 4. All states are at equilibrium. The gas can be modeled as a closed system. Ignore changes of KE and PE. The following properties in the table fix the states: State 1 p (bar) 2 3 4 8 6 7 5 tz 3 2 1 Note: State 1 to 2 is a constant-temperature process. State 2-3 is a constant-volume process. State 3-4 is a constant-temperature process. Ol p (bar) 1 (a) Mark the states 1-4 and draw (roughly and manually) the three processes (1-2, 2-3, 3-4) on the following p-v diagram. 0 3 6 1 3 v (m³/kg) 3 1 1 2 2 300K T (K) 300 600K 300 3 4 5 v (m³/kg) 600 600 6 7 8 (b) Use equations and concise texts to explain and answer the following questions for the process 2-3. (i) How does the internal energy change during the process: no change, increase, or decrease (AU)? (ii) How does the enthalpy change during the process (AH)? (iii) Is the work negative, positive, or zero during the process (W)? (iv) Is the heat transfer negative, positive, or zero during the process (Q)? An ideal gas (the weight is m kg) in an idealized piston-cylinder assembly undergoes a series of processes from state 1 to states 2, 3 and 4. All states are at equilibrium. The gas can be modeled as a closed system. Ignore changes of KE and PE. The following properties in the table fix the states: State 1 p (bar) 2 3 4 8 6 7 5 tz 3 2 1 Note: State 1 to 2 is a constant-temperature process. State 2-3 is a constant-volume process. State 3-4 is a constant-temperature process. Ol p (bar) 1 (a) Mark the states 1-4 and draw (roughly and manually) the three processes (1-2, 2-3, 3-4) on the following p-v diagram. 0 3 6 1 3 v (m³/kg) 3 1 1 2 2 300K T (K) 300 600K 300 3 4 5 v (m³/kg) 600 600 6 7 8 (b) Use equations and concise texts to explain and answer the following questions for the process 2-3. (i) How does the internal energy change during the process: no change, increase, or decrease (AU)? (ii) How does the enthalpy change during the process (AH)? (iii) Is the work negative, positive, or zero during the process (W)? (iv) Is the heat transfer negative, positive, or zero during the process (Q)?
Expert Answer:
Related Book For 

Business Communication Essentials a skill based approach
ISBN: 978-0132971324
6th edition
Authors: Courtland L. Bovee, John V. Thill
Posted Date:
Students also viewed these general management questions
-
They are seen as time-consuming, demotivating, inaccurate, biased, and unfair. A McKinsey survey indicates most CEOs don't find the appraisal process in their companies helps to identify top...
-
The PV diagram in Fig. 15-23 shows two possible states of a system containing 1.35 moles of a monatomic ideal gas, (P1 = P2 = 455 N/m2, V1 = 2.00 m3, V2 = 8.00m3.) (a) Draw the process which depicts...
-
(Multiple choice) (1) The temperature change of two blocks of masses MA and MB is the same when they absorb equal amounts of heat. It follows that the specific heats are related by (a) cA =...
-
Different theories about early childhood inform approaches to children's learning and development. Early childhood educators draw upon a range of perspectives in their work ..." (EYLF p.12)....
-
Refer to the information for Presidio, Inc., in PA5-4. Additional information for Presidio's most recent year of operations follows: Number of units...
-
The billing cycle goes from May 1 to May 31. The balance at the start of the billing cycle is \(\$ 450.21\). The list of transactions on the card is below. Find the average daily balance for the...
-
What problems arise when transfer pricing is used?
-
One of the biggest problems of student writers is paraphrasing secondary sources correctly to avoid plagiarism. Your Task. For each of the following, read the original passage. Analyze the...
-
Requirement 2: The company has just hired a new marketing manager who insists that unit sales can be dramatically increased by dropping the selling price from $8 to $7. The marketing manager would...
-
Provide the missing information. (Always use cell references and formulas where appropriate to receive full credit. If you copy/paste from the Instructions tab you will be marked wrong.) Item Case 1...
-
How are prepaid expenses, accrued expenses, unearned revenue, and accrued revenue similar to each other? a. Prepaid expenses and accrued expenses are both reported as expenses in the income statement...
-
James just bought a 7% annual coupon bond that will mature in 10 years for $960. Assume the par value is $1,000. What is the yield-to-maturity of this bond? Disney has a semi-annual 7% coupon bond....
-
Suppose a government has a deficit, and the government finances its deficit by issuing bonds. a. Do you think that it may have any impact on the Monetary Base of the economy? Explain your answer. b....
-
Advertisers can mislead through exaggeration by making claims unsupported by evidence. Puffery is the supposedly harmless use of superlatives and subjective praise in advertisements. Give an example...
-
Suppose the TLK Corporation issued bonds a few years ago with a par value of $1,000 and an annual interest payment of $130. If their current yield is 10 percent, their current market (or resale)...
-
Provide an assessment of the likely effectiveness of the Federal Government's policies and actions in assisting companies withstand the negative impact of the effective shutdown of many economic...
-
Discrete Disorder Select a DSM disorder (in case utilize 299.00 Autism Spectrum Disorder) and, for the sake of argument, formulate a way to understand that disorder that assumes it is not continuous,...
-
At 31 December 20X9, the end of the annual reporting period, the accounts of Huron Company showed the following: a. Sales revenue for 20X9, $ 2,950,000, of which one- quarter was on credit. b....
-
A not-so-secret secret is getting more attention than you'd really like after an article in Business week gave the world an inside look at how much money you and other electronics retailers make from...
-
What are the six traits of professionalism?
-
Read the following document and (a) Analyze the strengths and weaknesses of each sentence and (b) Revise the message so that it follows this chapter's guidelines. I am responding to your letter of...
-
Is outsourcing by U.S. firms to foreign countries beneficial to the U.S. economy? Weigh the pros and cons, and offer your conclusions.
-
a. The dollar is presently worth .8 euros. What is the direct exchange rate of the euro? b. The direct exchange rate of the euro is presently valued higher than it was last month. What does this...
-
Assume that the Polish currency (called zloty) is worth $.32. The U.S. dollar is worth .7 euros. A U.S. dollar can be exchanged for 8 Mexican pesos. Last year a dollar was valued at 2.9 Polish zloty,...

Study smarter with the SolutionInn App



