
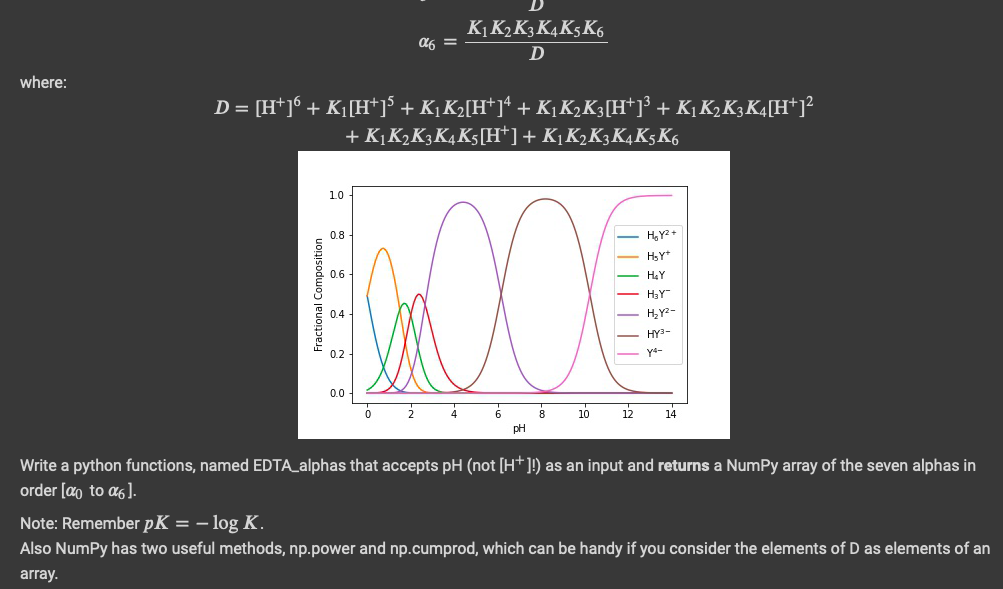
EDTA has many forms in an aqueous solution. The short hand notation for these forms, in order of decreasing protonation, is H4Y2+, H,Y+, H4Y, H3Y,H2Y2-, HY3-and Y4- do = ai with pK1 = 0.0, pK2 = 1.5, pK3 = 2.0, pK4 = 2.66, pK5= 6.16, pK6 = 10.24. At any pH a mass balance on EDTA requires that its total concentration equal the combined concentrations of each of its forms. CEDTA = [H, Y2+] + [HzY+] + [H4Y] + [H3Y-] + [H_Y2-] + [HY3-] + [Y-41 The fraction of EDTA in the form Y4- is: ya- = [Y4-1 CEDTA The relative concentrations of these species is pH dependent, and can be calculated with the alpha values for each species: [H+]6 D Ki[H+] D KK2[H"]4 a2 = D K1K2K3 [HT] al3 = D KK2K3K4[H"] 04 = D K1K2K3K4K5 [H"] as = D KKKK KK 26 = D where: KKK3K4KSK 26 = D where: D= [H+j + K1 [H+] + KK2[H+]4 + KK2 K3 [H+]} + K1 K2 K3K4[H+]? + KiK,K3K4 K5 [H+] + KiKK3K4K5K6 10 0.8 H,Y2+ HsY 0.6 HY Fractional Composition w 0.4 HY H2Y2- HY- 0.2 74- 0.0 4 6 8 10 12 14 PH Write a python functions, named EDTA_alphas that accepts pH (not [H+]!) as an input and returns a NumPy array of the seven alphas in order [ao to ab]. Note: Remember pK = - log K. Also NumPy has two useful methods, np.power and np.cumprod, which can be handy if you consider the elements of D as elements of an array. EDTA has many forms in an aqueous solution. The short hand notation for these forms, in order of decreasing protonation, is H4Y2+, H,Y+, H4Y, H3Y,H2Y2-, HY3-and Y4- do = ai with pK1 = 0.0, pK2 = 1.5, pK3 = 2.0, pK4 = 2.66, pK5= 6.16, pK6 = 10.24. At any pH a mass balance on EDTA requires that its total concentration equal the combined concentrations of each of its forms. CEDTA = [H, Y2+] + [HzY+] + [H4Y] + [H3Y-] + [H_Y2-] + [HY3-] + [Y-41 The fraction of EDTA in the form Y4- is: ya- = [Y4-1 CEDTA The relative concentrations of these species is pH dependent, and can be calculated with the alpha values for each species: [H+]6 D Ki[H+] D KK2[H"]4 a2 = D K1K2K3 [HT] al3 = D KK2K3K4[H"] 04 = D K1K2K3K4K5 [H"] as = D KKKK KK 26 = D where: KKK3K4KSK 26 = D where: D= [H+j + K1 [H+] + KK2[H+]4 + KK2 K3 [H+]} + K1 K2 K3K4[H+]? + KiK,K3K4 K5 [H+] + KiKK3K4K5K6 10 0.8 H,Y2+ HsY 0.6 HY Fractional Composition w 0.4 HY H2Y2- HY- 0.2 74- 0.0 4 6 8 10 12 14 PH Write a python functions, named EDTA_alphas that accepts pH (not [H+]!) as an input and returns a NumPy array of the seven alphas in order [ao to ab]. Note: Remember pK = - log K. Also NumPy has two useful methods, np.power and np.cumprod, which can be handy if you consider the elements of D as elements of an array








