Answered step by step
Verified Expert Solution
Question
1 Approved Answer
G youtubeet-Google.. (17) YouTube 601 Espanola St SE slitherio Official Naruto MM.. DANH SCH ANIM ParentVUE Google Dich Eye Doctor Albu O KINETICS AND
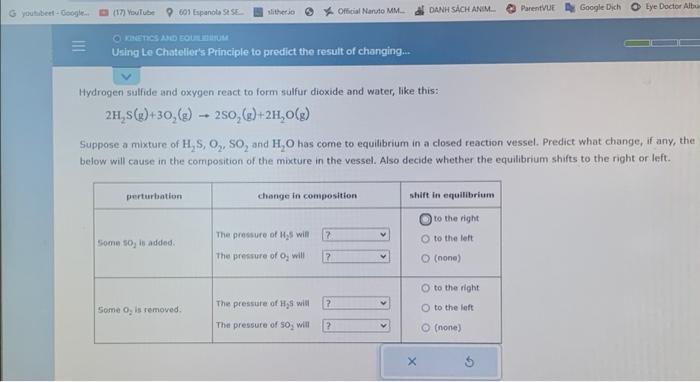
G youtubeet-Google.. (17) YouTube 601 Espanola St SE slitherio Official Naruto MM.. DANH SCH ANIM ParentVUE Google Dich Eye Doctor Albu O KINETICS AND EQUILIUM Using Le Chatelier's Principle to predict the result of changing... Hydrogen sulfide and oxygen react to form sulfur dioxide and water, like this: 2H,S(g)+30,(g) 250,(g)+2H,O(g) Suppose a mixture of H,S, O,, SO, and H,O has come to equilibrium in a closed reaction vessel. Predict what change, if any, the below will cause in the composition of the mixture in the vessel. Also decide whether the equilibrium shifts to the right or left. perturbation change in composition : shift in equilibrium to the right The pressure of H5 will 7 Some so, is added. O to the left The pressure of O, will ? (none) O to the right Some O, is removed. The pressure of HS will The pressure of So, will ? O to the left ? O (none) X
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


