Answered step by step
Verified Expert Solution
Question
1 Approved Answer
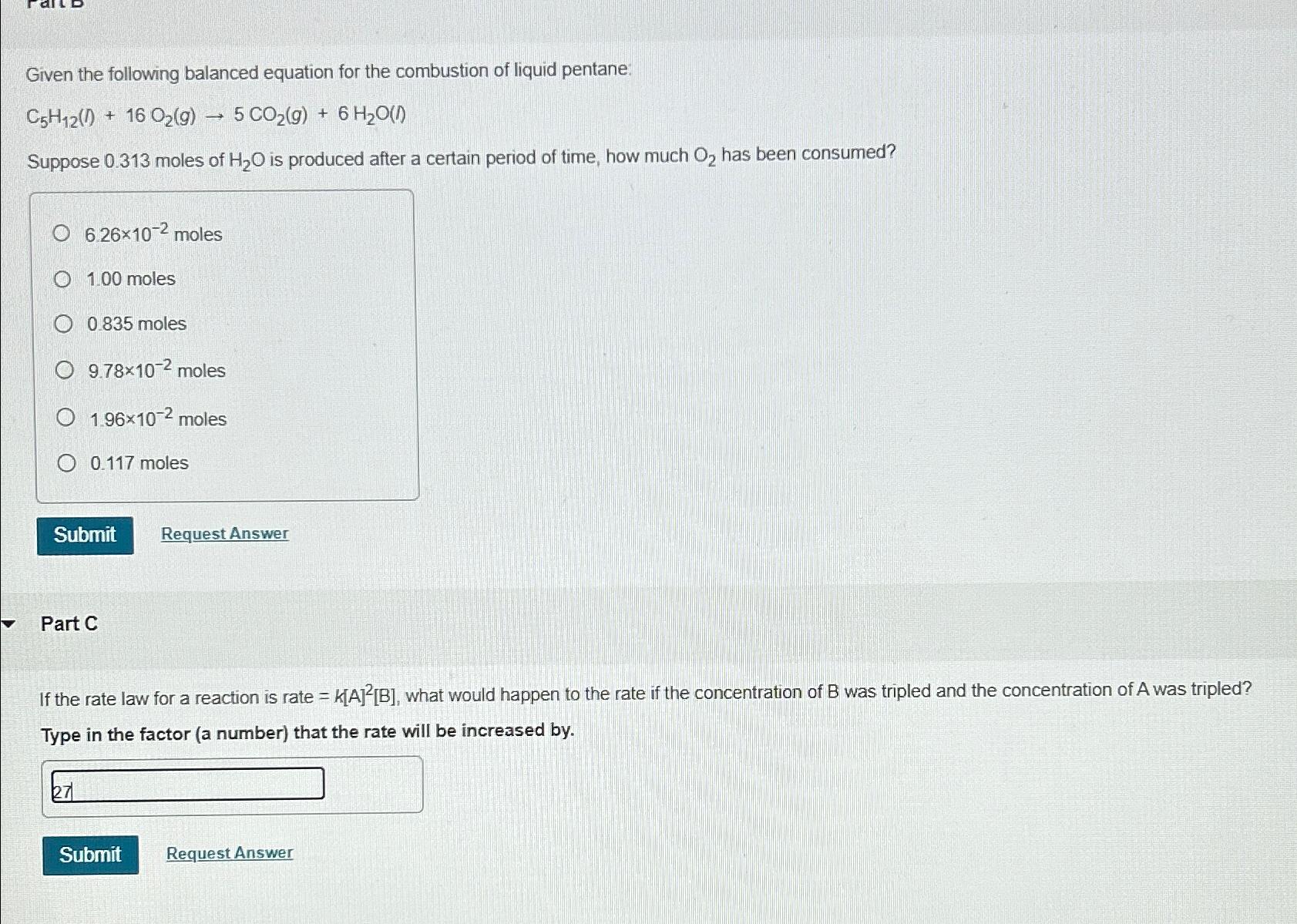
Given the following balanced equation for the combustion of liquid pentane: C 5 H 1 2 ( I ) + 1 6 O 2 (
Given the following balanced equation for the combustion of liquid pentane:
Suppose moles of is produced after a certain period of time, how much has been consumed?
moles
moles
moles
moles
moles
moles
Request Answer
Part C
If the rate law for a reaction is rate what would happen to the rate if the concentration of was tripled and the concentration of A was tripled? Type in the factor a number that the rate will be increased by
Request Answer
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


