Question
Given the graph of y = f(x) below, answer all of the following questions. -5 1 y 5 Activa (a) List the intervals where
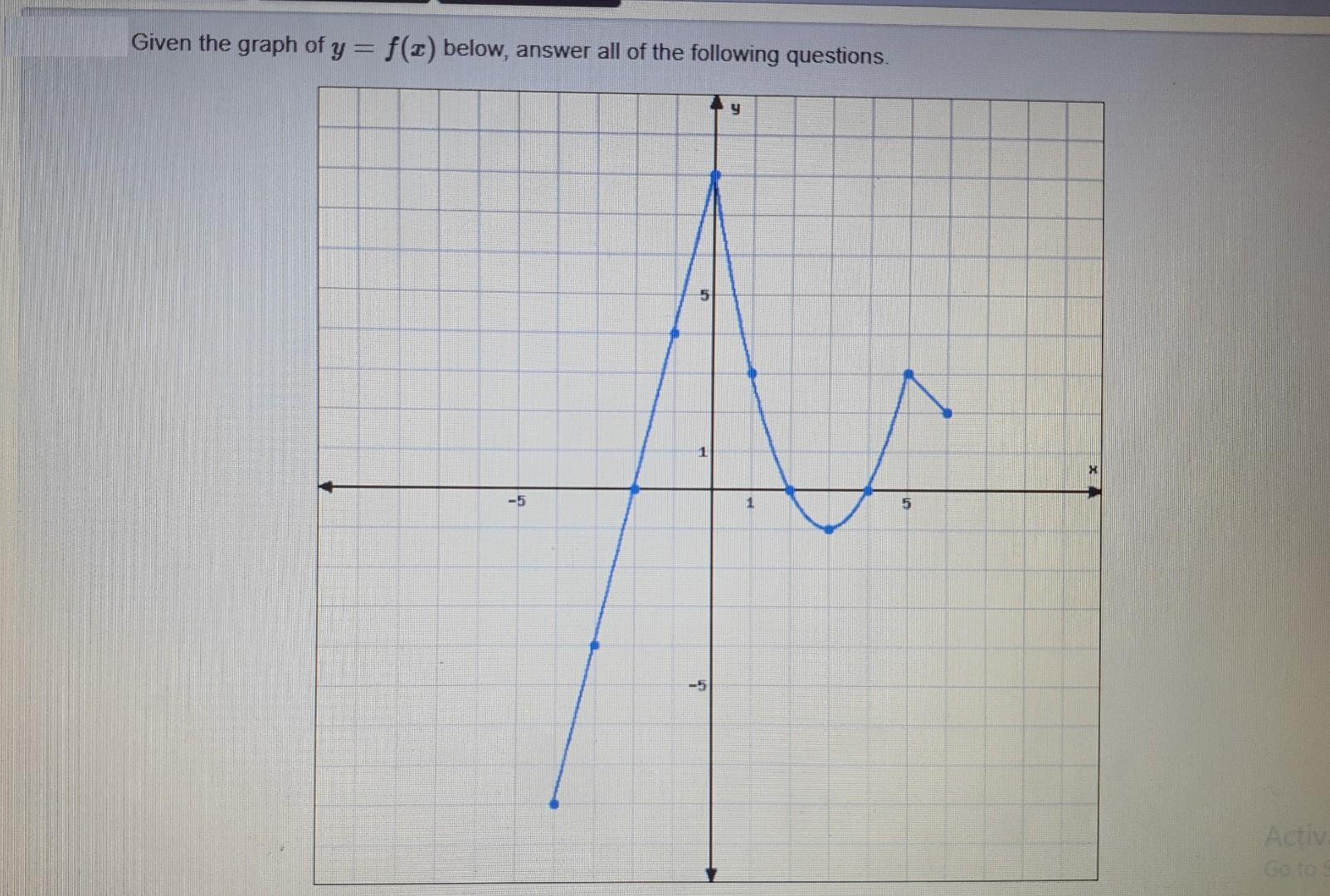

Given the graph of y = f(x) below, answer all of the following questions. -5 1 y 5 Activa (a) List the intervals where f is increasing: (b) List the intervals where f is decreasing: (c) List the distinct y-values of the local maximums, if any exist: (d) List the distinct y-values of the local minimums, if any exist: (e) Find the y-value of the maximum, if it exists: (f) Find the y-value of the minimum, if it exists:
Step by Step Solution
3.45 Rating (152 Votes )
There are 3 Steps involved in it
Step: 1
To answer the given questions we first need to determine where the function is increasing and decrea...
Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get StartedRecommended Textbook for
Digital Signal Processing
Authors: Jonh G. Proakis, Dimitris G.Manolakis
3rd Edition
978-0133737622, 133737624, 978-013373762
Students also viewed these Accounting questions
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
View Answer in SolutionInn App



