Answered step by step
Verified Expert Solution
Question
1 Approved Answer
help A solution contains 4.4g of urea (molar mass =60.06g/mol ), a nonvolatile compound, dissolved in 50.0g of water (molar mass-18.015 g/mol ). If the
help 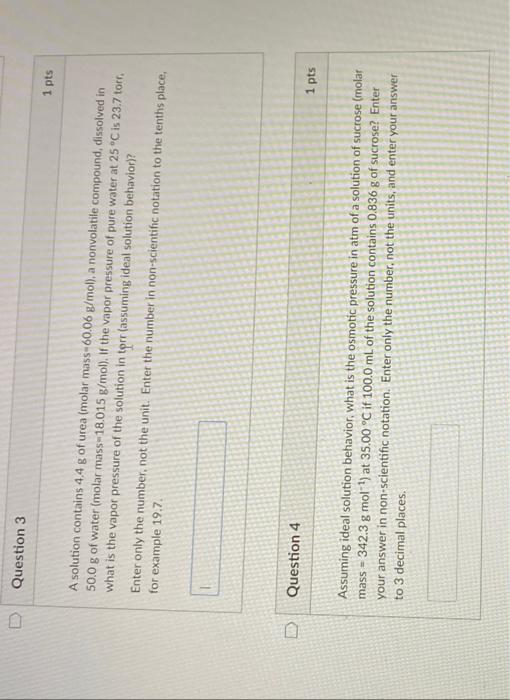
A solution contains 4.4g of urea (molar mass =60.06g/mol ), a nonvolatile compound, dissolved in 50.0g of water (molar mass-18.015 g/mol ). If the vapor pressure of pure water at 25C is 23.7 torr. what is the vapor pressure of the solution in tprr (assuming ideal solution behavior)? Enter only the number, not the unit. Enter the number in non-scientific notation to the tenths place, for example 19.7. Question 4 Assuming ideal solution behavior, what is the osmotic pressure in atm of a solution of sucrose (molar mass =342.3gmol1 ) at 35.00C if 100.0mL of the solution contains 0.836g of sucrose? Enter your answer in non-scientific notation. Enter only the number. not the units, and enter your answer to 3 decimal places 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


