help me find the unknown and flowcharts
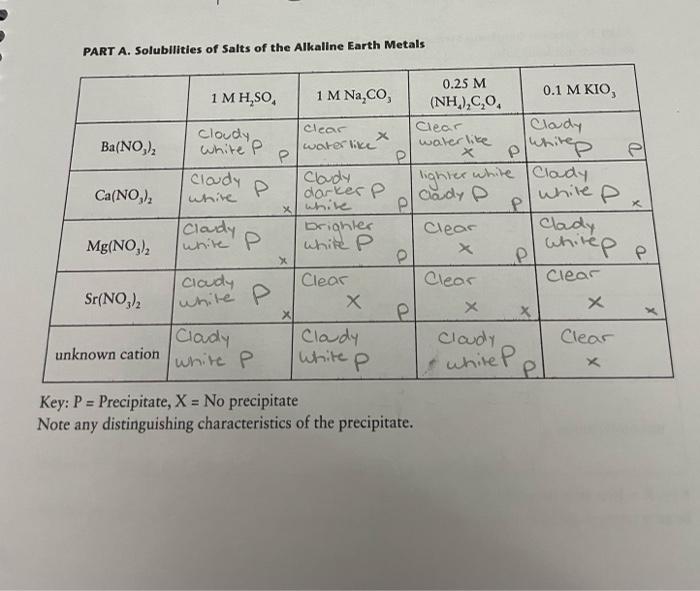
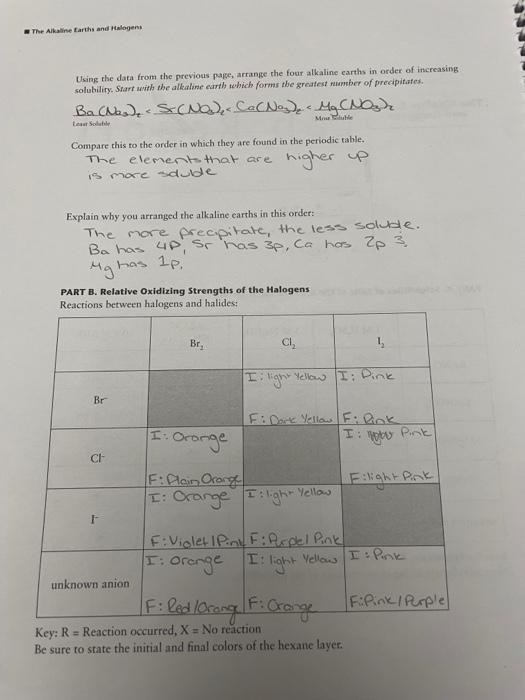
PART A. Solubilities of Salts of the Alkaline Earth Metals el x x white white P whitep 0.25 M 1 MH,SO 1 M Na,co, 0.1 M KIO, (NH4)2C.0, clear Clear Cloudy Cloudy X Ba(NO), White P water like water like P x Cloudy Cloudy white Clady lighter P Ca(NO) white darker P Claudy P while P P Cloudy brightes Clear Clady Mg(NO) white P P P ciady Clear Clear Clear Sr(NO) X X X P X Clady Clady Cloudy Clear unknown cation while P White P el Key: P = Precipitate, X = No precipitate Note any distinguishing characteristics of the precipitate. P X white P while P The Alkaline Earth and Halogen Using the data from the previous page, arrange the four alkaline earths in order of increasing solubility. Start with the alkaline earth which forms the greatest number of precipitates Ba(Nasd S. CND Co CNo Mg CNOgr. More Eltele Compare this to the order in which they are found in the periodic table. The element that are is more duble higher up Explain why you arranged the alkaline earths in this order: The more precpitate, the less soluble. Ba has up, sr has 3p, ca has 2p 3 Mg has le PART B. Relative Oxidizing Strengths of the Halogens Reactions between halogens and halides: Br, CI, 1 I light Yellow I Dink Br I Gronge F: Dante Yellow F: link I gobe Pink CH F:Plain Orange I: Crange I light Flight Pink Yellow IF: Violet Pink F: Ropel Pink I: Orange I light yellow I: Pink unknown anion F FPink Purple Key: R = Reaction occurred, X = No reaction Be sure to state the initial and final colors of the hexane layer. F: led lorong. Fi Crange Experiment Based on the data from the previous page, arrange the halogens in order of increasing oxidizing strength: Iz Weakest Serg Compare this to the order in which they are found in the periodic table. The Weakest (I) is lower on the periodic table and has more protocol nations. C. the strongest is higher on the periodic table has less protored rections PART C. Identification of an Unknown Alkaline Earth Hallde Flowchart of procedure you used for identifying the alkaline carth cation: Ba? Ca?o, Mg Sri. 2 To B Flowchart of procedure you used for identifying the halide ion: Unknown Number: #4 Identity of anion (X-) present: Identity of cation (M) present: Questions 1. In the reaction Cl +2Br2C+Br,, which species (CI, or Br) is the oxidizing agent? Cik 2. Which periodic group of metals is the most reactive? alkali metals 3. Name the following: C0, xalale Br Bromine I Todide