Answered step by step
Verified Expert Solution
Question
1 Approved Answer
henry's law application henry's law applications 6.2 6.3 Dimethyl ether + water at 473 K: 1.834 PIMPa) 1.765 2.165 0.003 0.002 3.185 0.012 3.654 2.498
henry's law application 

henry's law applications 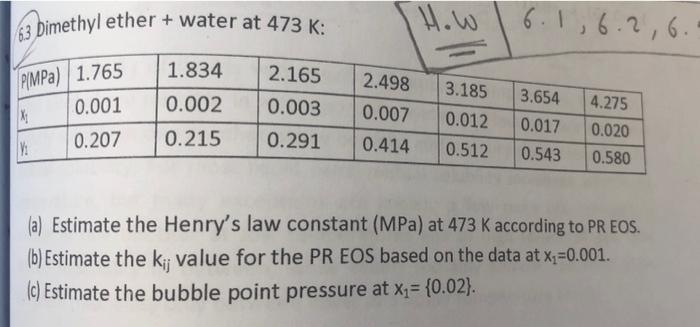
6.2 6.3 Dimethyl ether + water at 473 K: 1.834 PIMPa) 1.765 2.165 0.003 0.002 3.185 0.012 3.654 2.498 0.007 0.414 0.001 0.207 X; 0.215 0.291 4.275 0.020 0.580 0.017 0.543 0.512 63 Dimethyl ether + water at 473 K: How 6.1,6.26. 2.165 PlMPa) 1.765 1.834 0.002 2.498 3.185 0.001 0.003 M 0.012 3.654 0.017 0.543 0.007 0.414 0.215 4.275 0.020 0.580 0.207 0.291 0.512 (a) Estimate the Henry's law constant (MPa) at 473 K according to PREOS. (b) Estimate the ki, value for the PR EOS based on the data at x=0.001. (c) Estimate the bubble point pressure at x = {0.02). 6.2 6.3 Dimethyl ether + water at 473 K: 1.834 PIMPa) 1.765 2.165 0.003 0.002 3.185 0.012 3.654 2.498 0.007 0.414 0.001 0.207 X; 0.215 0.291 4.275 0.020 0.580 0.017 0.543 0.512 63 Dimethyl ether + water at 473 K: How 6.1,6.26. 2.165 PlMPa) 1.765 1.834 0.002 2.498 3.185 0.001 0.003 M 0.012 3.654 0.017 0.543 0.007 0.414 0.215 4.275 0.020 0.580 0.207 0.291 0.512 (a) Estimate the Henry's law constant (MPa) at 473 K according to PREOS. (b) Estimate the ki, value for the PR EOS based on the data at x=0.001. (c) Estimate the bubble point pressure at x = {0.02) 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


