Answered step by step
Verified Expert Solution
Question
1 Approved Answer
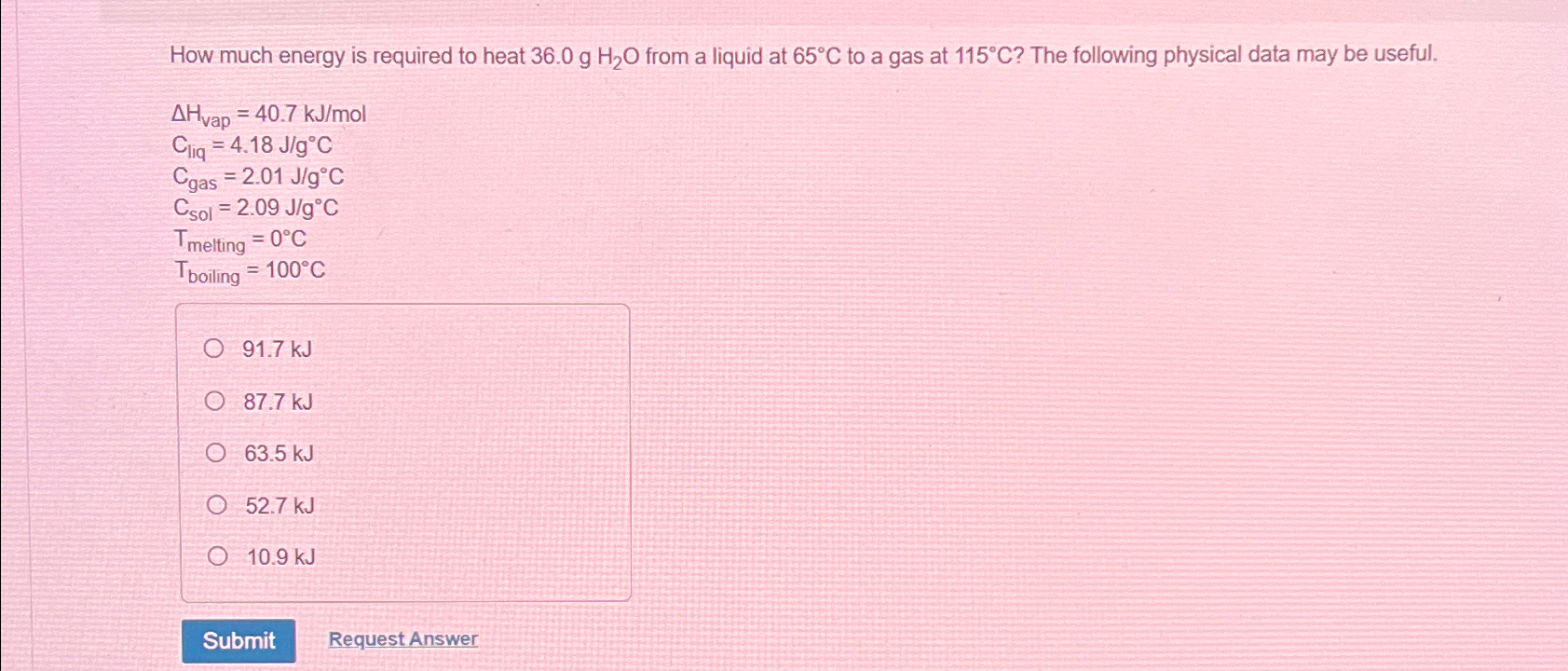
How much energy is required to heat 36.0gHH_(2)O from a liquid at 65deg C to a gas at 115deg C ? The following physical data
How much energy is required to heat
36.0gHH_(2)Ofrom a liquid at
65\\\\deg Cto a gas at
115\\\\deg C? The following physical data may be useful.\
\\\\Delta H_(vap )=40.7k(J)/(m)ol\ C_(liq )=4.18(J)/(g)\\\\deg C\ C_(gas )=2.01(J)/(g)\\\\deg C\ C_(sol )=2.09(J)/(g)\\\\deg C\ T_(melting )=0\\\\deg C\ T_(boiling )=100\\\\deg C\
91.7kJ\
87.7kJ\
63.5kJ\
52.7kJ\
10.9kJ\ Request Answer
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


