Answered step by step
Verified Expert Solution
Question
1 Approved Answer
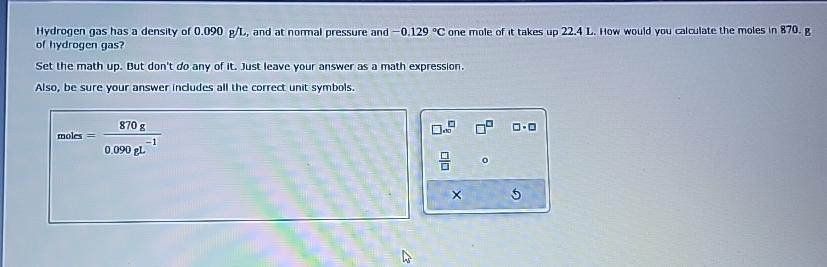
Hydrogen gas has a density of 0 . 0 9 0 g L , and at normal pressure and - 0 . 1 2 9
Hydrogen gas has a density of and at normal pressure and one mole of it takes up How would you calculate the moles in g of hydrogen gas?
Set the math up But don't do any of it Just leave your answer as a math expression.
Also, be sure your answer includes all the correct unit symbols.
moles
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


