Answered step by step
Verified Expert Solution
Question
1 Approved Answer
i have included the correct answers as a reference if you need it for the last part. please only solve the system temperature and pressure
i have included the correct answers as a reference if you need it for the last part. please only solve the system temperature and pressure part 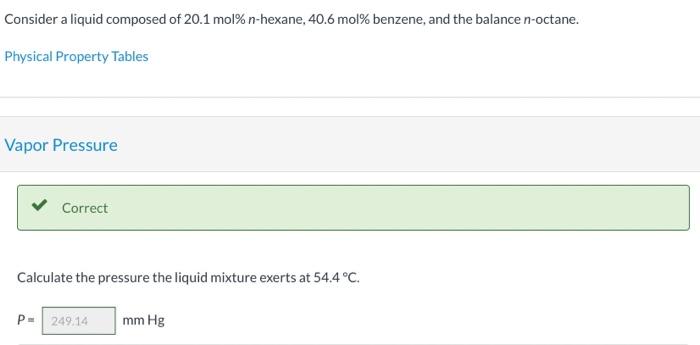
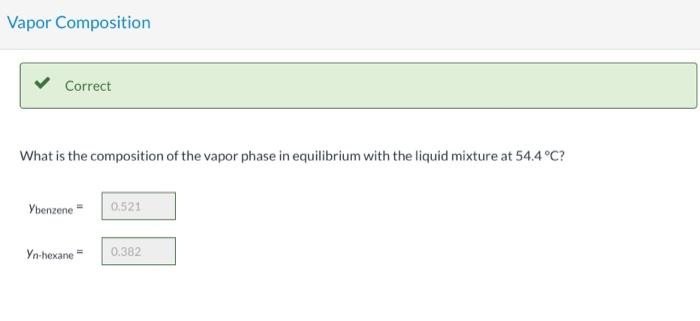
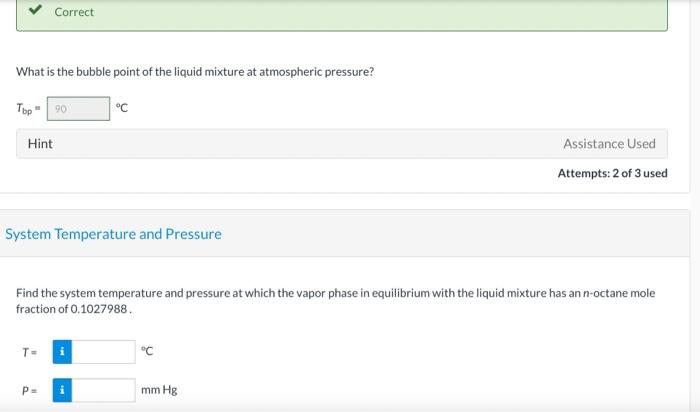
Consider a liquid composed of 20.1 mol% n-hexane, 40.6 mol% benzene, and the balance n-octane. Physical Property Tables Vapor Pressure Correct Calculate the pressure the liquid mixture exerts at 54.4C. P249.14 mm Hg Vapor Composition Correct What is the composition of the vapor phase in equilibrium with the liquid mixture at 54.4C? Ybenzene 0,521 Yn-hexane 0.382 Correct What is the bubble point of the liquid mixture at atmospheric pressure? Top 20 C Hint Assistance Used Attempts: 2 of 3 used System Temperature and Pressure Find the system temperature and pressure at which the vapor phase in equilibrium with the liquid mixture has an n-octane mole fraction of 0.1027988 T- i C PE mm Hg 


Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


