Answered step by step
Verified Expert Solution
Question
1 Approved Answer
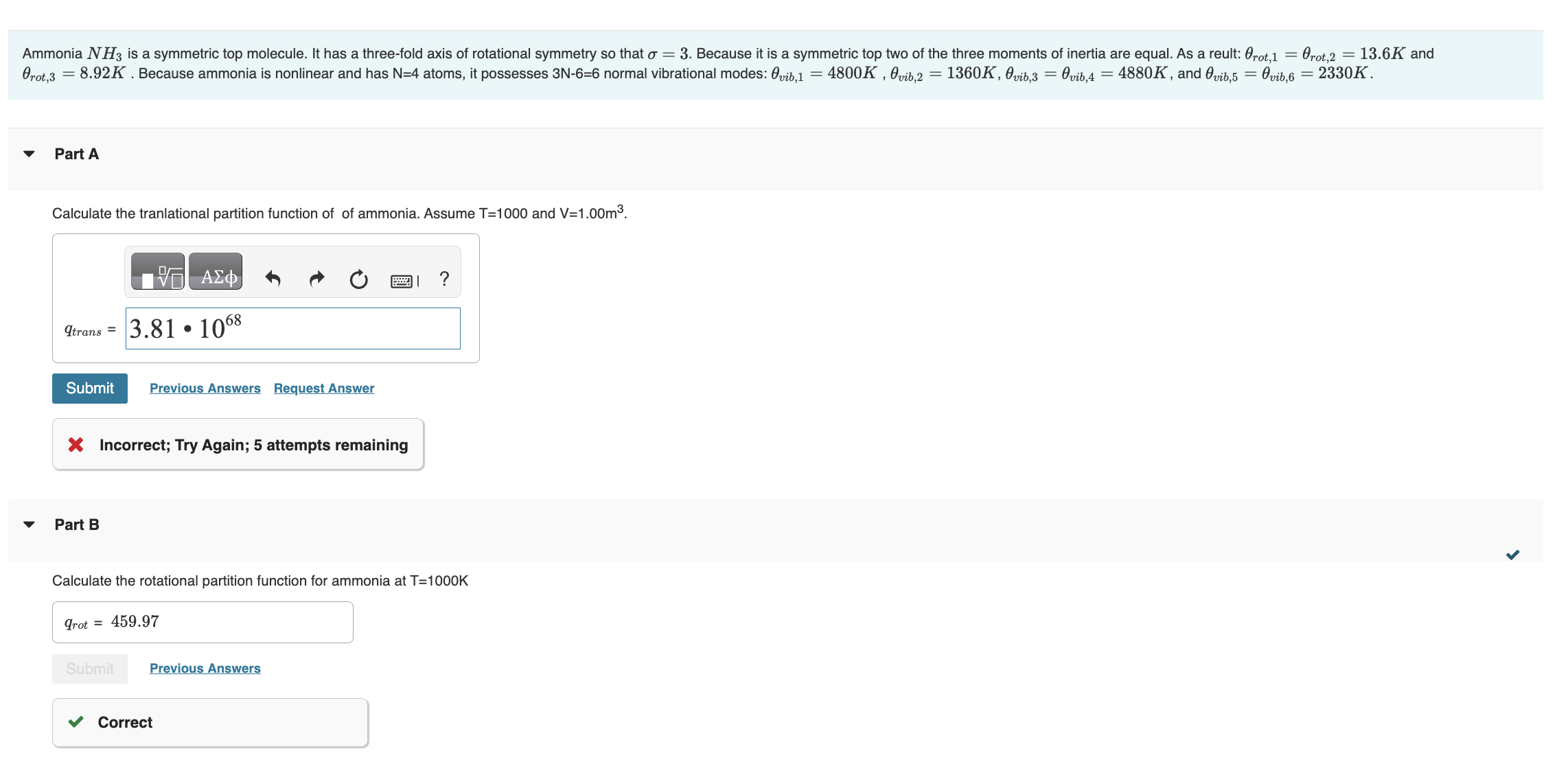
I NEED HELP WITH A, F & G PLEASE! Part A Calculate the tranlational partition function of ammonia. Assume T=1000 and V=1.00m3. Incorrect; Try Again;
I NEED HELP WITH A, F & G PLEASE!
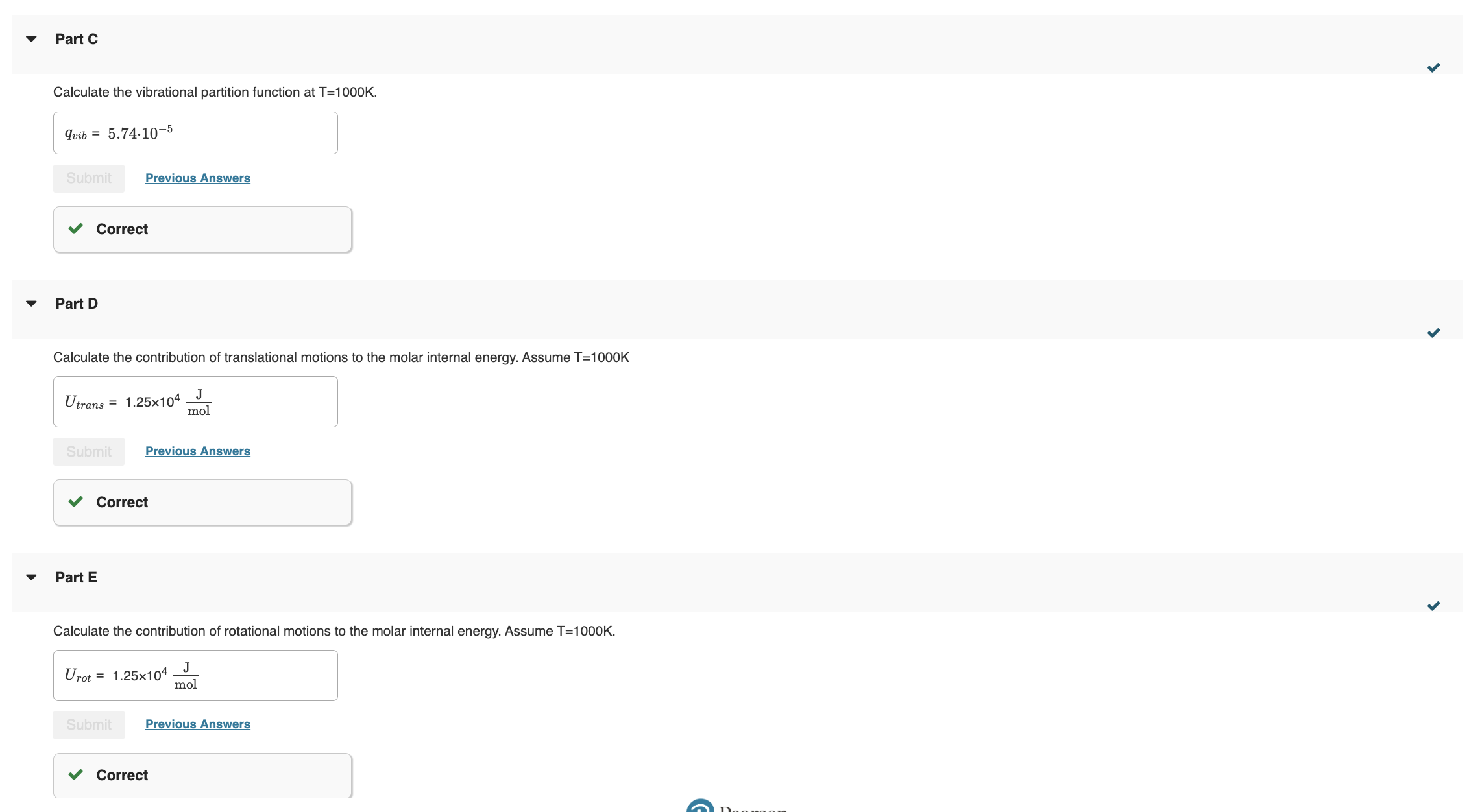
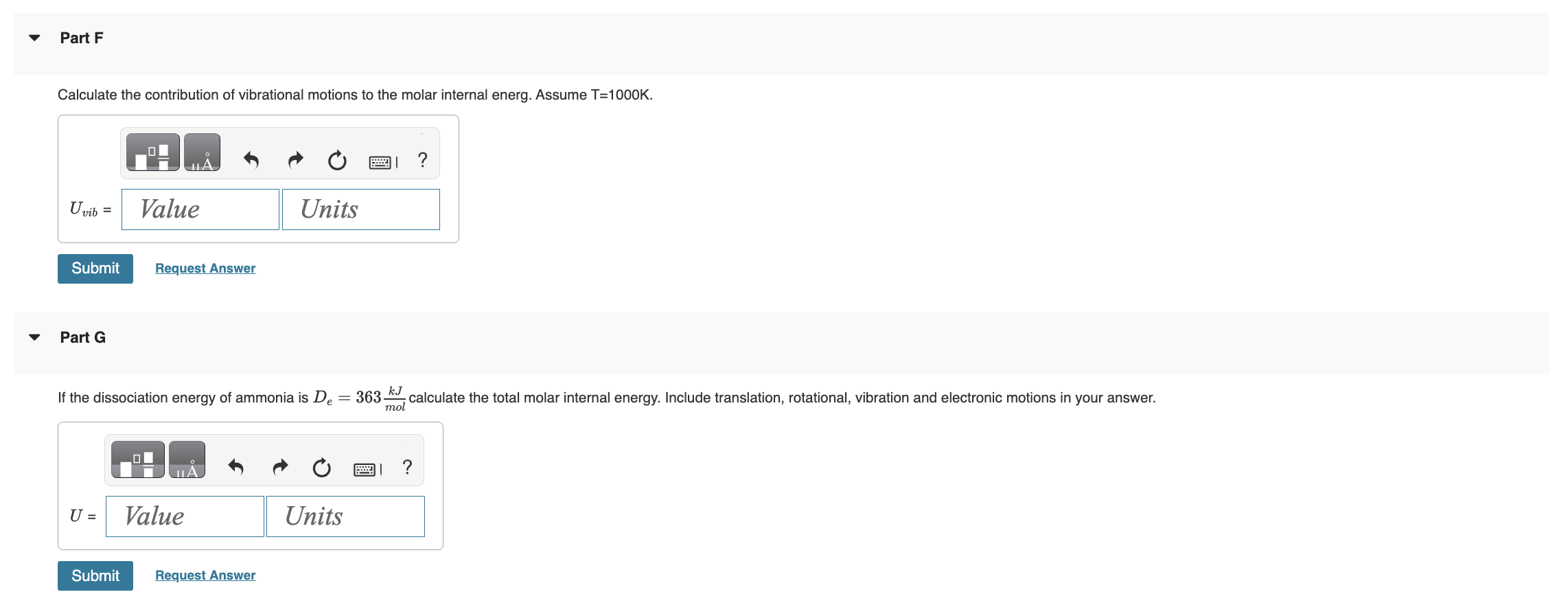
Part A Calculate the tranlational partition function of ammonia. Assume T=1000 and V=1.00m3. Incorrect; Try Again; 5 attempts remaining Part B Calculate the rotational partition function for ammonia at T=1000K Calculate the vibrational partition function at T=1000K. Part D Calculate the contribution of translational motions to the molar internal energy. Assume T=1000K Part E Calculate the contribution of rotational motions to the molar internal energy. Assume T=1000K. Calculate the contribution of vibrational motions to the molar internal energ. Assume T=1000K. Part G If the dissociation energy of ammonia is De=363molkJ calculate the total molar internal energy. Part A Calculate the tranlational partition function of ammonia. Assume T=1000 and V=1.00m3. Incorrect; Try Again; 5 attempts remaining Part B Calculate the rotational partition function for ammonia at T=1000K Calculate the vibrational partition function at T=1000K. Part D Calculate the contribution of translational motions to the molar internal energy. Assume T=1000K Part E Calculate the contribution of rotational motions to the molar internal energy. Assume T=1000K. Calculate the contribution of vibrational motions to the molar internal energ. Assume T=1000K. Part G If the dissociation energy of ammonia is De=363molkJ calculate the total molar internal energyStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


