Answered step by step
Verified Expert Solution
Question
1 Approved Answer
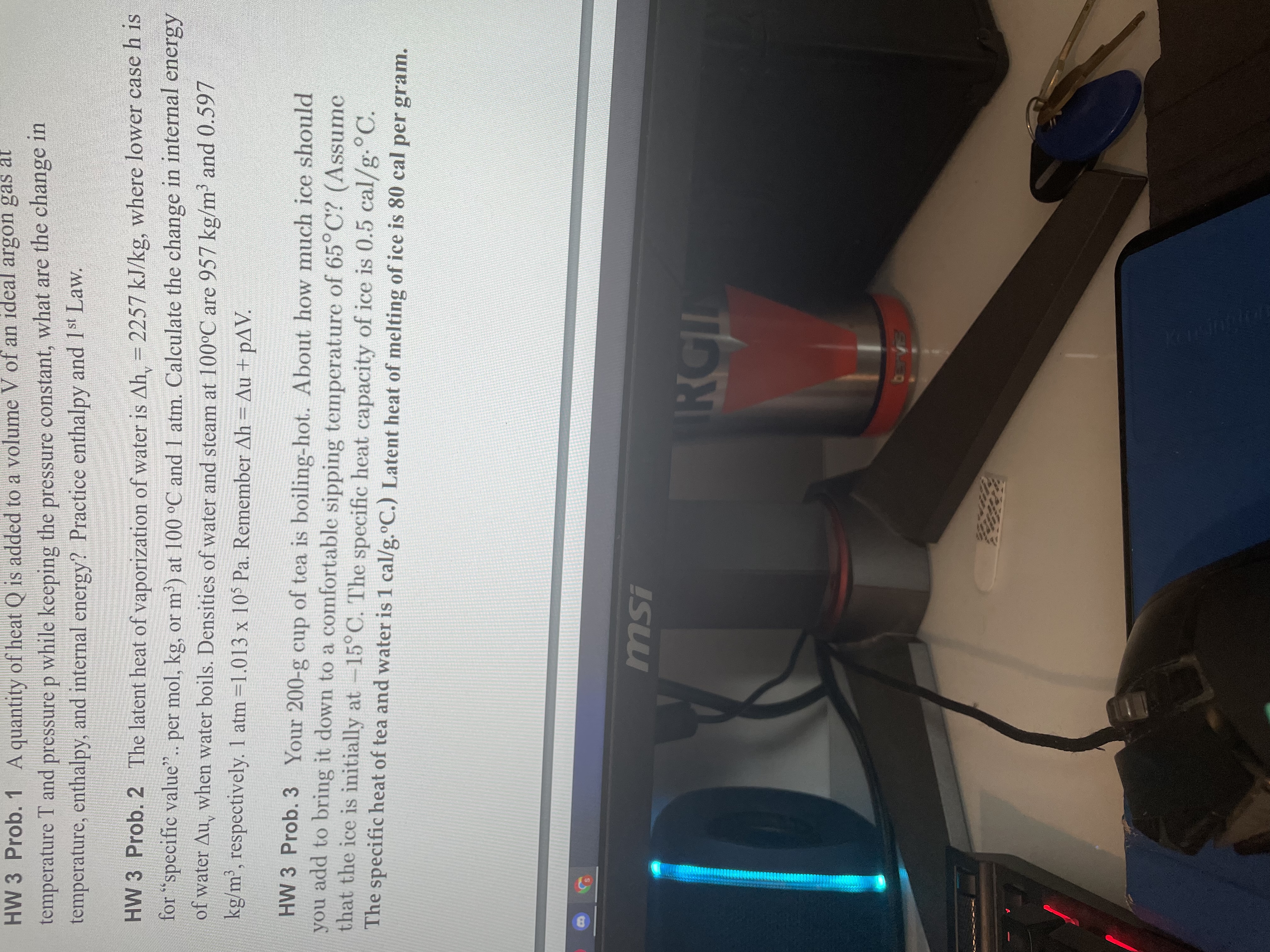
I'm only stuck on the first two. For the first one I know how to do the version of this problem when it is done
I'm only stuck on the first two. For the first one I know how to do the version of this problem when it is done at a constant volume because the amount of work is zero. I am trying to use that same approach but nothing here is equal to zero because work is being done. So I am having trouble seeing how to relate these three quantities. And for the second one, I do not know how to apply the problem given since p is not given
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


