Answered step by step
Verified Expert Solution
Question
1 Approved Answer
in organic reaction how you can know that all the reactants have completely reacted with each other, you can find this out when you do
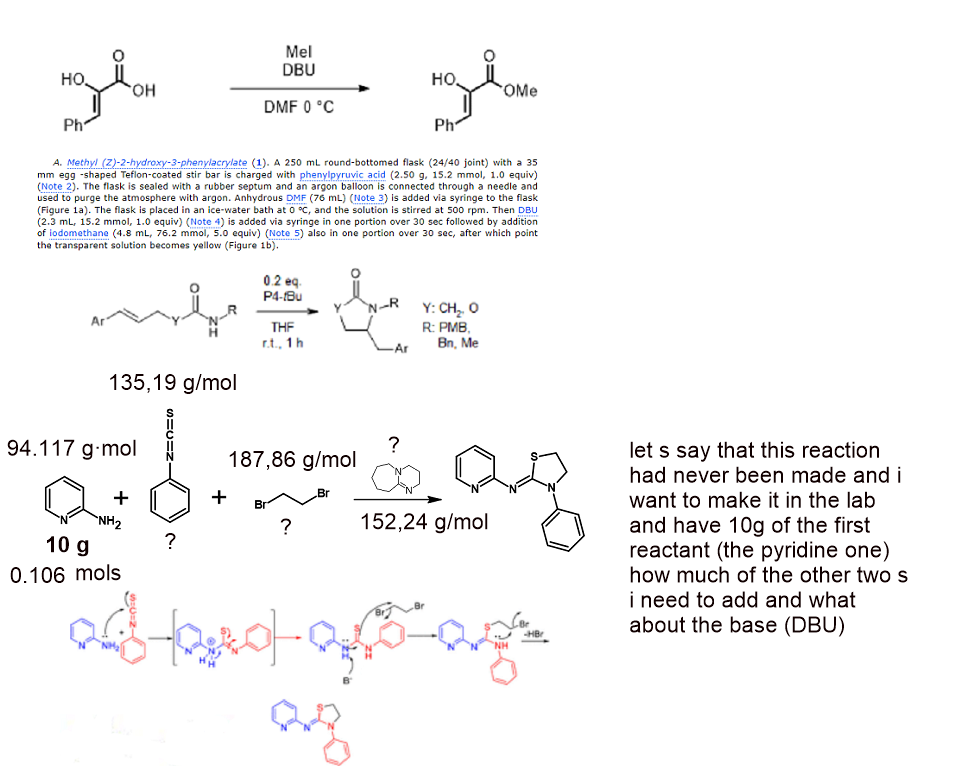
in organic reaction how you can know that all the reactants have completely reacted with each other, you can find this out when you do the chemical synthesis and analyze what happened in the flask with different methods and then adjust the weights of the reactants so that nothing is wasted or it can be on paper, I know how to convert grams to mmol and to find out what is the limiting reagent and then to find out the equivalents the weight ratio between the reagents but one thing cannot be understood by me and I want very much. If for example, I invent a chemical reaction that has never been done before and I draw the mechanism on paper, and after seeing that it is possible, I want to do it in real life, and I have g of the first reactant, where can I find out how much to add from the others so that the reaction should be completed completely, I understood the idea of the molar mass, but what I want is to explain how the chemists know that the iodomethane reagent must be added as an equivalent of if they know the quantity of only the first compound or this matter, as I said above, is also analyzed in the laboratory directly and determine the most stable weights?
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


