In which of the following sets do all species have the same number of electrons? +...
Fantastic news! We've Found the answer you've been seeking!
Question:
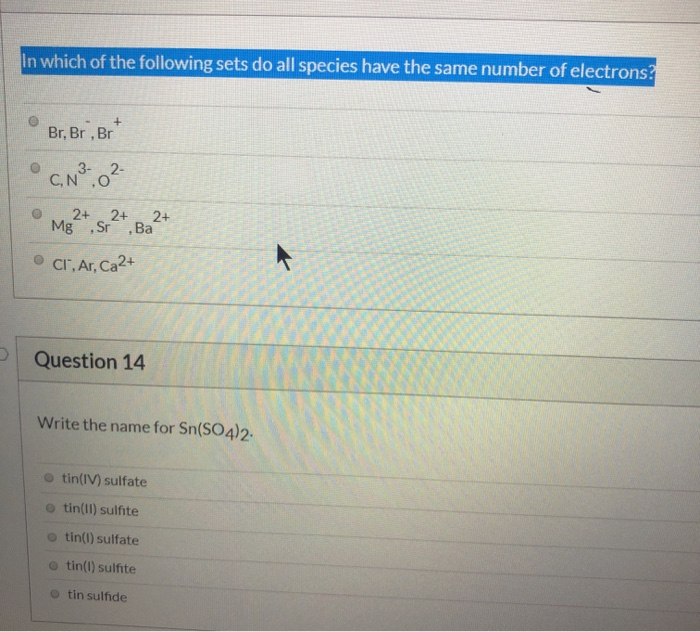
Transcribed Image Text:
In which of the following sets do all species have the same number of electrons? + Br, Br, Br 3- C.N³.0²- 2- 2+ 2+ 2+ Ba Mg Sr CI", Ar, Ca²+ Question 14 Write the name for Sn(SO4)2. tin(IV) sulfate tin(II) sulfite tin(1) sulfate tin(1) sulfite Otin sulfide In which of the following sets do all species have the same number of electrons? + Br, Br, Br 3- C.N³.0²- 2- 2+ 2+ 2+ Ba Mg Sr CI", Ar, Ca²+ Question 14 Write the name for Sn(SO4)2. tin(IV) sulfate tin(II) sulfite tin(1) sulfate tin(1) sulfite Otin sulfide
Expert Answer:
Answer rating: 100% (QA)
Answer G option D is correct species having same no of elections are called A Br 8 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which of the following sets of quantum numbers are not allowed in the hydrogen atom? For the sets of quantum numbers that are incorrect, state what is wrong in each set. a. n = 3, = 2, m = 2 b. n =...
-
Which of the following sets of quantum numbers are not allowed? For each incorrect set, state why it is incorrect. a. n = 3, = 3, m = 0, ms = - 1/2 b. n = 4, = 3, m = 2, ms = - 1/2 c. n = 4, = 1,...
-
Which of the following sets of circumstances require a government to treat another entity as a component unit of its reporting entity? Why? 1. The government appoints 3 of the 7 members of the...
-
1. If the nominal policy interest rate is 5% and the expected rate of inflation is 3%, what is the value for the vertical intercept of the LM curve? 2. If the expected rate of inflation were to...
-
Hope Hospital purchased a special X-ray machine. The machine, which cost $311,560, was expected to last ten years, with an estimated residual value of $31,560. After two years of operation (and...
-
Movie companies need to predict the gross receipts of individual movies after a movie has debuted. The following results, listed in PotterMovies, are the first weekend gross, the U.S. gross, and the...
-
What is 83 modulo 7 ?
-
Presented below is information related to Blowfish radios for Hootie Company for the month of July. Instructions (a) Assuming that the periodic inventory method is used, compute the inventory cost at...
-
Medina Builders plans to sell 15-year, $1,000 face value zerocoupon bonds. The bonds is priced at $322. Assuming semi-annualcompounding, the yield to maturity for this bond isA. 7.7%B. 8.9%C. 12. 2...
-
3. Alpha Investors On July 1, 1985, Jim Roberts felt certain the stock market was in for a correction. The S&P 500 Index had risen 14% since the first of the year, and most analysts were predicting...
-
A lender estimates that the closing costs on a $170,000 home loan will be $6,375.00. The actual closing costs were 4.0% of the loan amount. Determine if the closing costs were higher or lower than...
-
On her way to work Tuesday, February 26, Ann Forrester ducked her head against the cold wind and stepped gingerly across the ice patches on Highway 328 to put a letter in the mailbox. Her husband,...
-
A narrow beam of monochromatic light enters diamond from air at an incident angle of 6 5 degrees. If the speed of light in air is 3 . 0 0 x 1 0 ^ 8 m / s and the speed of light in diamond is 1 . 2 4...
-
Croton Enterprises has worked out the activities, duration, and predecessors for its new project to create a new drink for the local Trinidad and Tobago market and for export to the Caribbean market....
-
What are the five major perspectives in the field of psychology. 1. Name all of the major perspectives (5 perspectives)? 2. Include the major theorists who influenced each perspective? 3. What...
-
Tommy Ltd. Sells t-shirts. The total sales for the week was $8,000 and the contribution margin was $0.96. The company sold 2,000 t-shirts for the week. Compute the variable cost per t-shirt .
-
Zdenka fast-tracked her studies and finished her program 4- months earlier than expected on February 1st. Her original completion date was June 1st and her study permit was issued until October 1st....
-
Keating & Partners is a law firm specializing in labour relations and employee-related work. It employs 25 professionals (5 partners and 20 managers) who work directly with its clients. The average...
-
What enone product would you except from aldol condensation of each of the followingcompounds? (c) CH2H H (b) -CH3
-
Draw the structure of cyclic adenosine mono phosphate (cAMP), a messenger involved in the regulation of glucose production in the body. Cyclic AMP has a phosphate ring connecting the 3' and 5'...
-
Rank the following compounds in order of their expected reactivity toward S N 2 reaction: CH 3 Br, CH 3 OTos, (CH 3 ) 3 CCl, (CH 3 ) 2 CHCl
-
Aim Inc. had the following activity for the years 2020-2022. - Prepaid maintenance contract: \(\$ 30,000\) on January 1,2020 , for a three-year period beginning January 1 , 2020. - Deferred revenue:...
-
On December 31, 2020, Colgait Inc. had an installment sale receivable balance of \(\$ 90,000\) recognized on its financial statements, while the amount was not recognized for tax purposes. Colgait...
-
Tyson Corporation reported pretax income from operations in 2020 of \(\$ 80,000\) (the first year of operations). In 2021, the corporation experienced a \(\$ 40,000\) NOL (pretax loss from...

Study smarter with the SolutionInn App


