Answered step by step
Verified Expert Solution
Question
1 Approved Answer
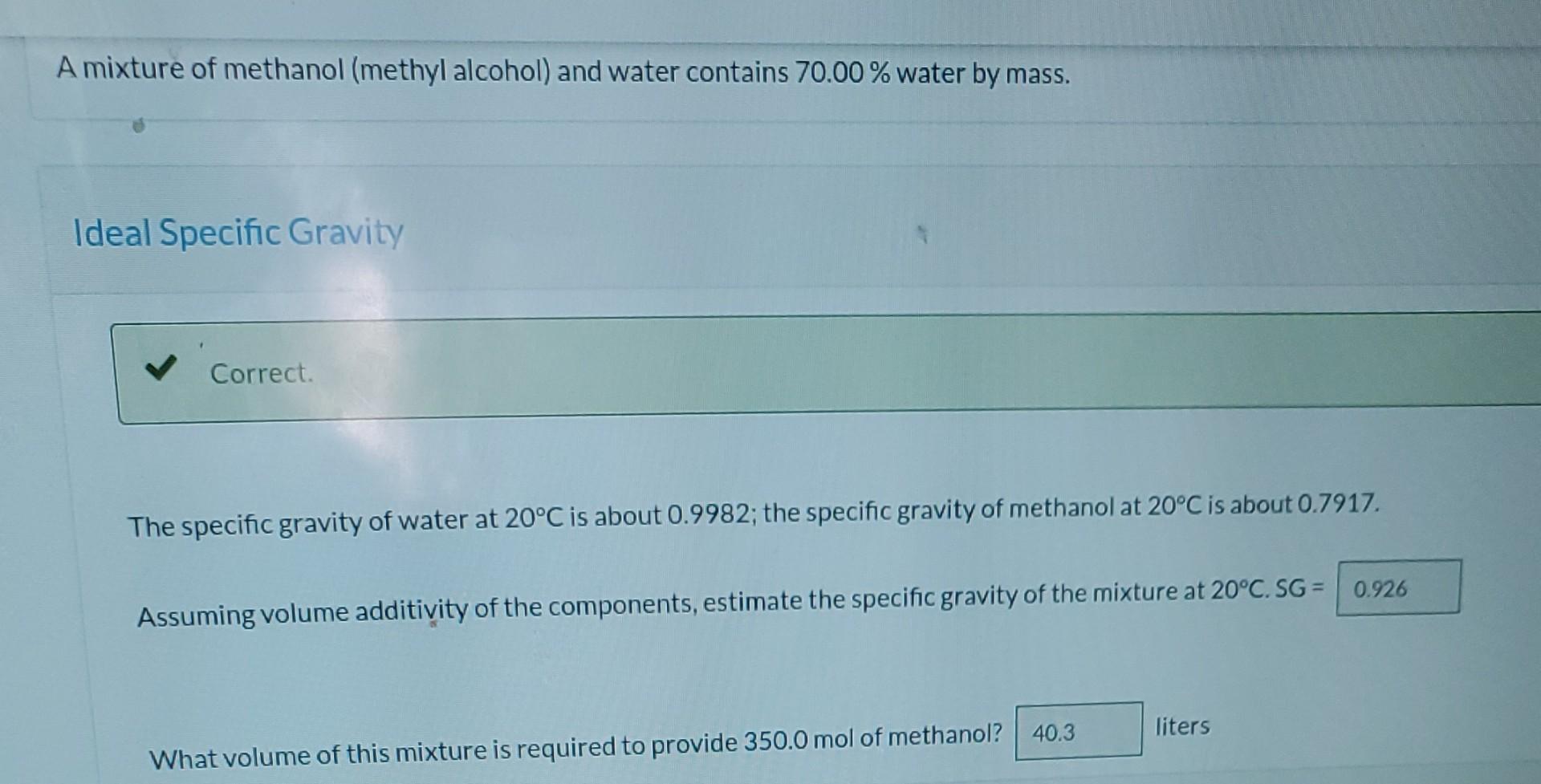
just the last 2 questions are needed please A mixture of methanol (methyl alcohol) and water contains 70.00% water by mass. Ideal Specific Gravity Correct.
just the last 2 questions are needed please
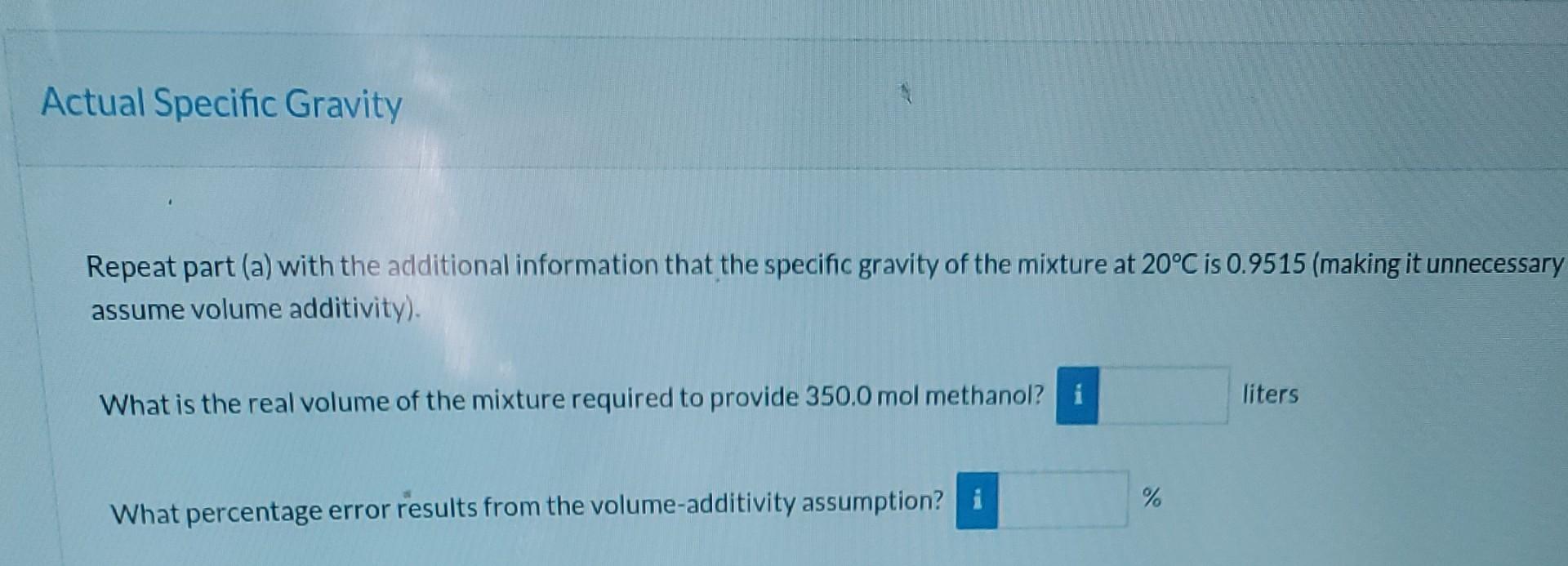
A mixture of methanol (methyl alcohol) and water contains 70.00% water by mass. Ideal Specific Gravity Correct. The specific gravity of water at 20C is about 0.9982; the specific gravity of methanol at 20C is about 0.7917. Assuming volume additivity of the components, estimate the specific gravity of the mixture at 20C. SG = 0.926 liters What volume of this mixture is required to provide 350.0 mol of methanol? 40.3 Actual Specific Gravity Repeat part (a) with the additional information that the specific gravity of the mixture at 20C is 0.9515 (making it unnecessary assume volume additivity). What is the real volume of the mixture required to provide 350.0 mol methanol? i liters % What percentage error results from the volume additivity assumptionStep by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


