Answered step by step
Verified Expert Solution
Question
1 Approved Answer
LiNiO2 is a cathode material for Li-ion batteries, a cheaper and more environmentally friendly alternative to LiCoO2. It has a layered structure with Li and
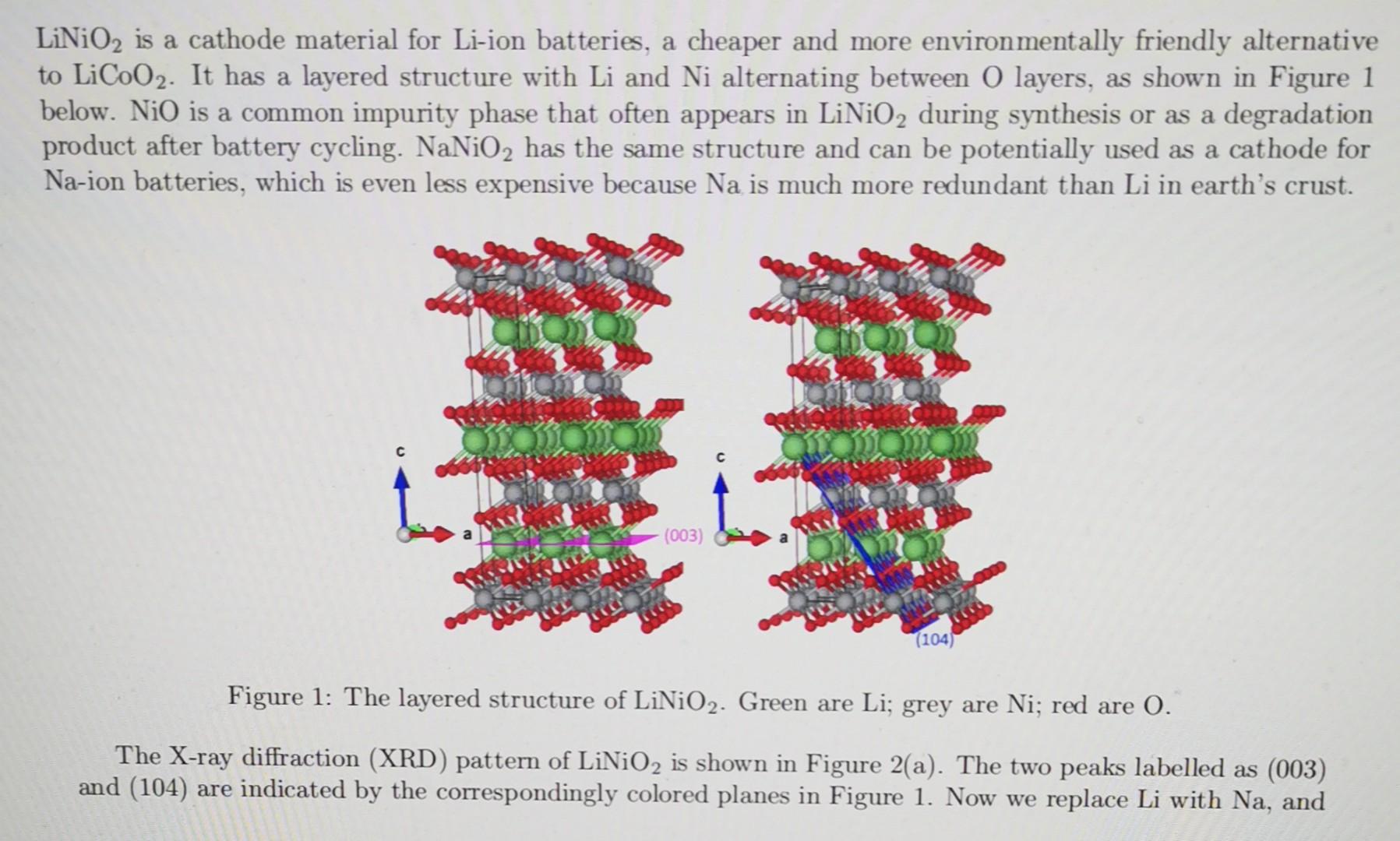

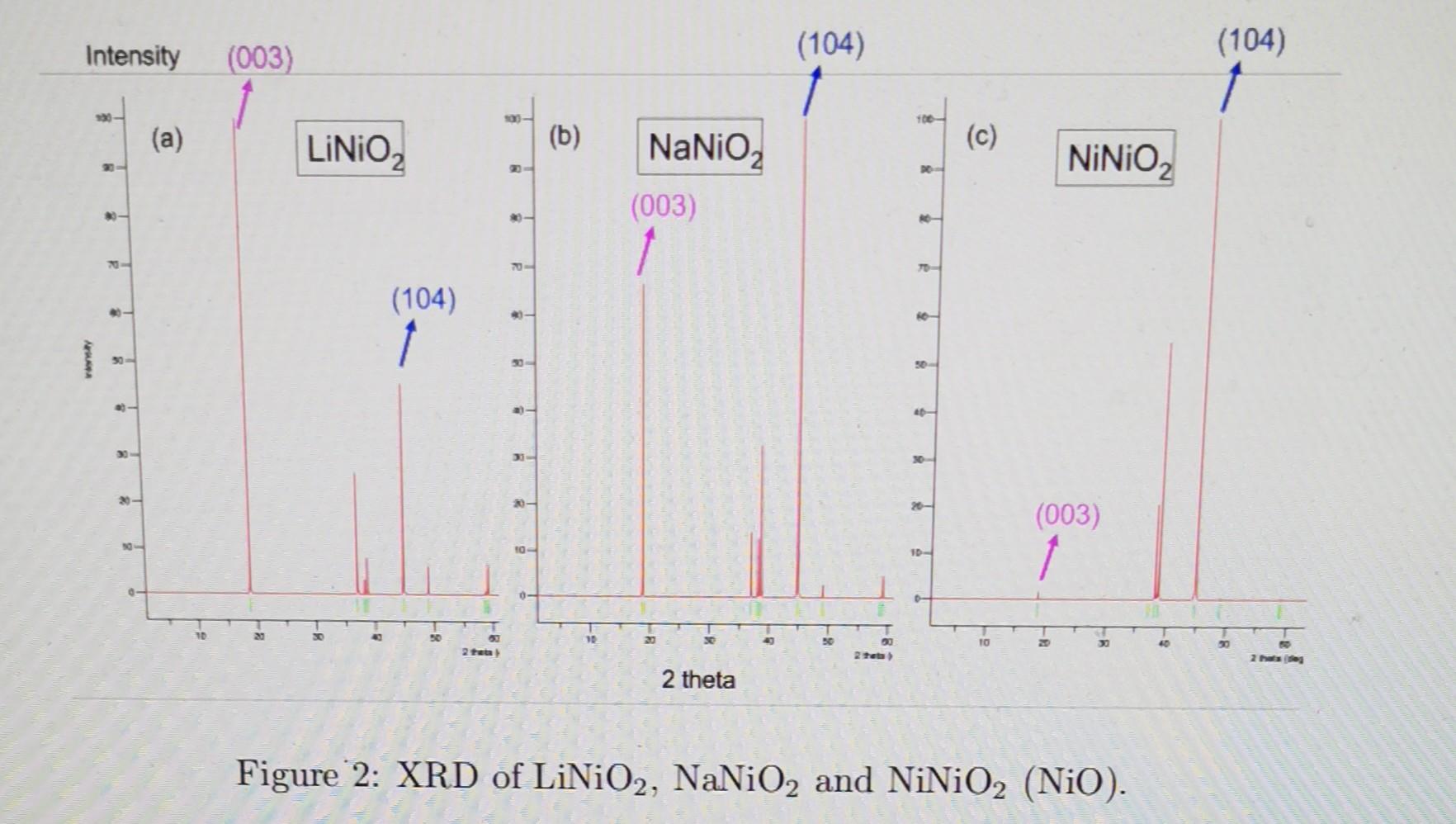
LiNiO2 is a cathode material for Li-ion batteries, a cheaper and more environmentally friendly alternative to LiCoO2. It has a layered structure with Li and Ni alternating between O layers, as shown in Figure 1 below. NiO is a common impurity phase that often appears in LiNiO2 during synthesis or as a degradation product after battery cycling. NaNiO2 has the same structure and can be potentially used as a cathode for Na-ion batteries, which is even less expensive because Na is much more redundant than Li in earth's crust. Figure 1: The layered structure of LiNiO2. Green are Li; grey are Ni; red are O. The X-ray diffraction (XRD) pattern of LiNiO2 is shown in Figure 2(a). The two peaks labelled as (003) and (104) are indicated by the correspondingly colored planes in Figure 1. Now we replace Li with Na, and the new XRD is shown in Figure 2(b). The obvious change is that the (003) peak becomes lower than the (104). Next we replace Li with Ni, and the XRD is shown in Figure 2(c). Could you give a qualitative explanation for this peak intensity trend as we go from Li to Na and then Ni? (Hint: you can simplify the structure to BCC, replace the center atom and calculate the structure factors) Figure 2: XRD of LiNiO2,NaNiO2 and NiNiO2(NiO)
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


