

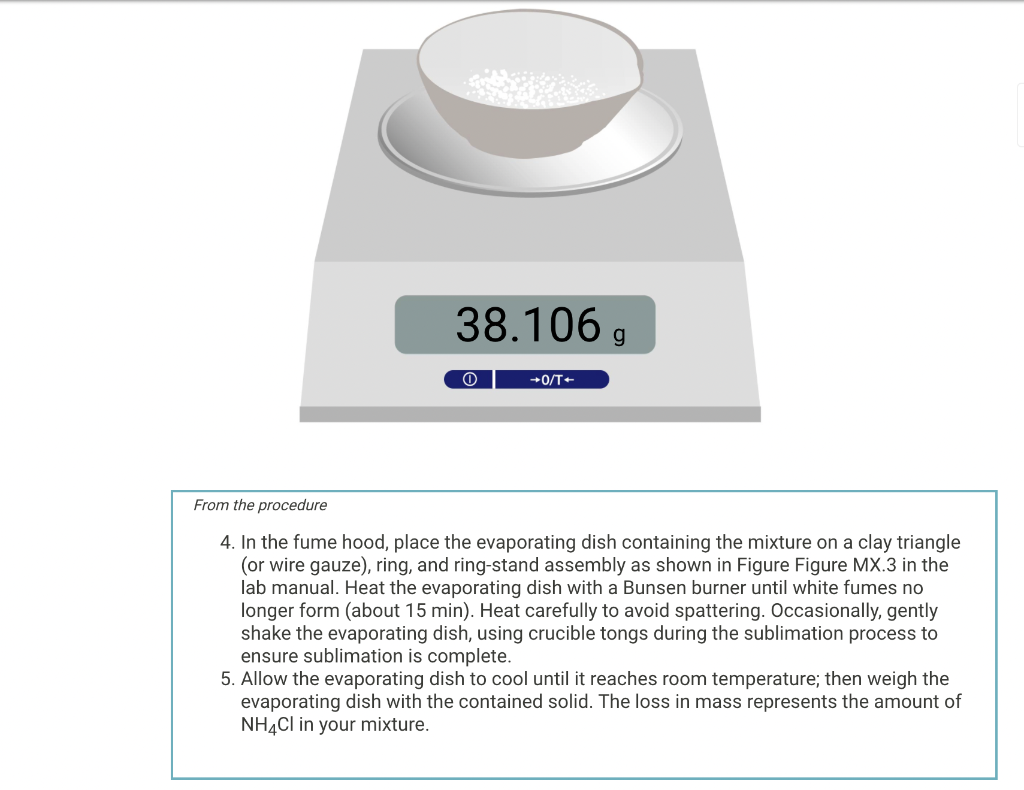




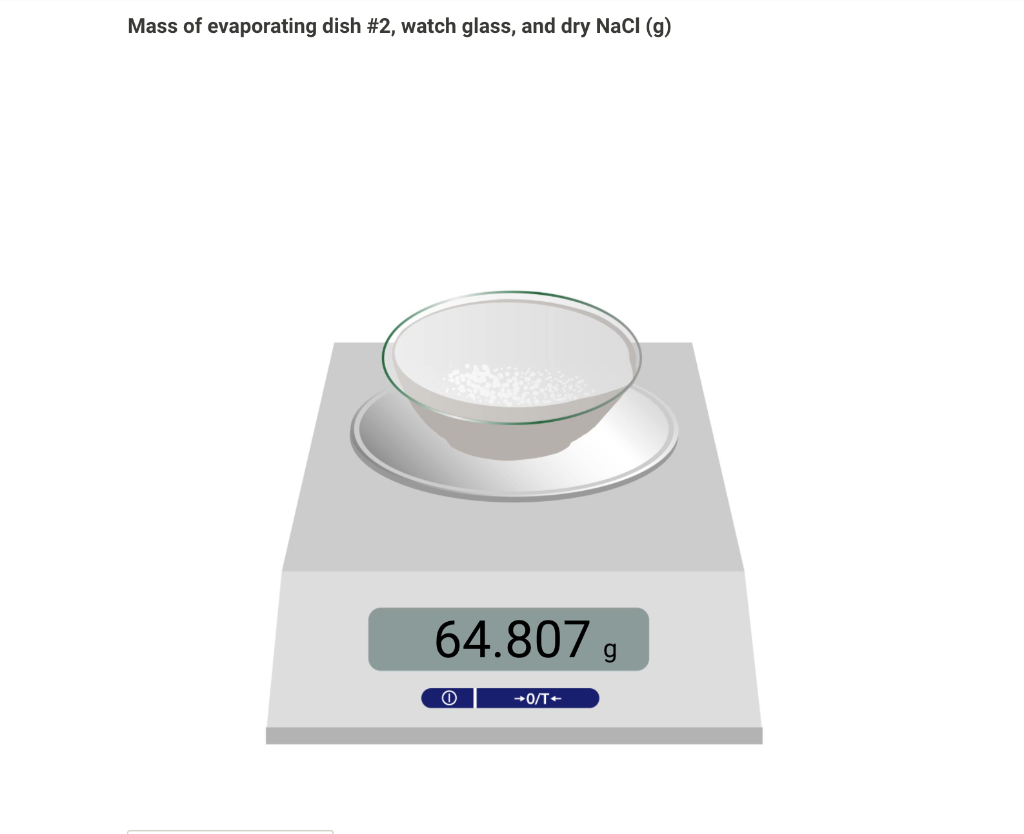
Mass of evaporating dish #1 (g) 37.150, O +0/T+ From the procedure 3. Obtain from your instructor a sample of the unknown mixture. Write the unknown number on your report sheet. If you obtain your unknown from a bottle, shake the bottle to make the sample mixture as uniform as possible. Add between 0.8 to 1.0 g of the sample to the evaporating dish, recording the exact sample mass to the nearest 0.001 g. You have been assigned the unknown number 3. Enter your unknown number: Mass of evaporating dish #1 and original sample (g) 38.106, 0 +0/T From the procedure 4. In the fume hood, place the evaporating dish containing the mixture on a clay triangle (or wire gauze), ring, and ring-stand assembly as shown in Figure Figure MX.3 in the lab manual. Heat the evaporating dish with a Bunsen burner until white fumes no longer form (about 15 min). Heat carefully to avoid spattering. Occasionally, gently shake the evaporating dish, using crucible tongs during the sublimation process to ensure sublimation is complete. 5. Allow the evaporating dish to cool until it reaches room temperature; then weigh the evaporating dish with the contained solid. The loss in mass represents the amount of NH4Cl in your mixture. Mass of evaporating dish #1 and sample after subliming NH4Cl (9) 37.986 , +0/T- From the procedure 6. Record the mass of evaporating dish #2 and separately, the mass of a watch glass to use as a lid Mass of evaporating dish #2 (g) 35.740 , Mass of watch glass (g) 28.705, 0 +0/T From the procedure 7. Measure 25 mL of Dl water in a graduated cylinder. 8. Remove the salt from the sand by adding small portions (2-3 mL) of Dl water (from the graduated cylinder) to the sand-salt mixture in evaporating dish #1. With a stirring rod gently, stir the mixture. Decant the liquid carefully into the evaporating dish #2, which you have weighed, being careful not to transfer any of the solid into evaporating dish #2. 9. Add more Dl water to the solid in evaporating dish #1, stir, and decant this liquid into evaporating dish #2 as before. Repeat the process a total of three times, using no more than 25 mL of Dl water. This process dissolves (extracts) the NaCl from the sand. You now have two evaporating dishes - one containing wet sand, #1, and the other containing a solution of sodium chloride, #2. 10. Carefully place evaporating dish #2, which contains the sodium chloride solution, on the clay triangle on the ring stand. Begin heating the solution gently to evaporate the water. Take care to avoid boiling or spattering, especially when liquid is present. 11. Near the end of the process, cover the evaporating dish with the watch glass that was weighed with evaporat-ing dish #2 and reduce the heat to prevent spattering. When you have dried the sodium chloride completely, no more water will condense on the watch glass and it too will be dry. 12. Let the evaporating dish and watch glass cool to room temperature on a wire gauze and weigh them. The difference between this mass and the mass of the empty evaporating dish and watch glass is the mass of the Naci. Mass of evaporating dish #2, watch glass, and dry NaCl (9) Mass of evaporating dish #2, watch glass, and dry NaCl (9) 64.807 g 0 +0/T














