Answered step by step
Verified Expert Solution
Question
1 Approved Answer
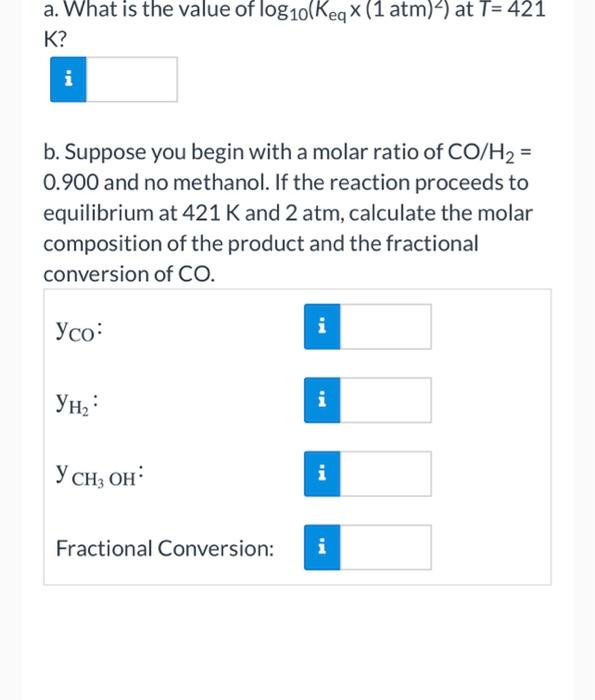
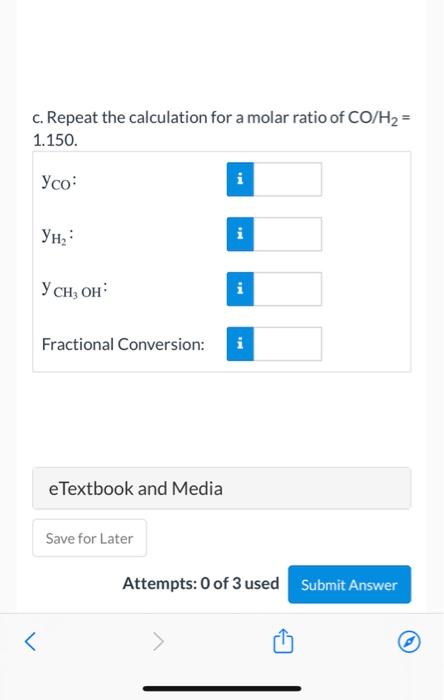
Methanol (C) is formed from carbon monoxide (A) and hydrogen (B) in the gas-phase reaction CO + 2H 2 CH 3 OH The mole fractions
Methanol (C) is formed from carbon monoxide (A) and hydrogen (B) in the gas-phase reaction
The mole fractions of the reactive species at equilibrium satisfy the relation
where P is the total pressure (atm), Keq the reaction equilibrium constant (atm2) , and T the temperature (K). The equilibrium constant Keq equals 10.5 at 373 K, and 2.316 x 104 at 573 K. A semilog plot of Keq(logarithmic scale) versus 1/T (rectangular scale) is approximately linear between T = 300 K and T = 600 K.
CO + 2H2 CH3OH
The mole fractions of the reactive species at equilibrium satisfy the relation
where P is the total pressure (atm), Keq the reaction equilibrium constant (atm2) , and T the temperature (K). The equilibrium constant Keq equals 10.5 at 373 K, and 2.316 x 104 at 573 K. A semilog plot of Keq(logarithmic scale) versus 1/T (rectangular scale) is approximately linear between T = 300 K and T = 600 K.
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


