Answered step by step
Verified Expert Solution
Question
1 Approved Answer
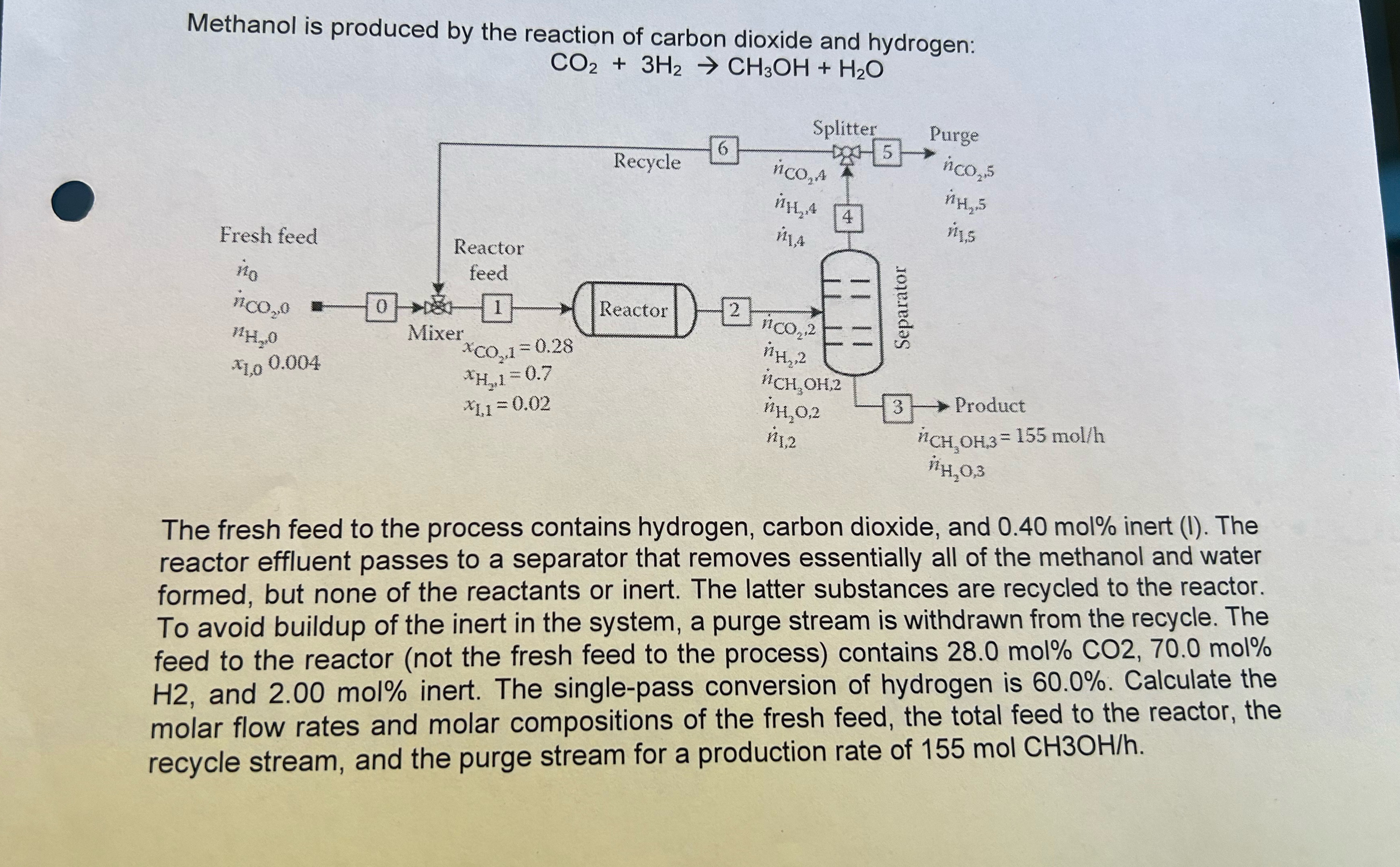
Methanol is produced by the reaction of carbon dioxide and hydrogen: C O 2 + 3 H 2 C H 3 O H + H
Methanol is produced by the reaction of carbon dioxide and hydrogen:
The fresh feed to the process contains hydrogen, carbon dioxide, and mol inert The reactor effluent passes to a separator that removes essentially all of the methanol and water formed, but none of the reactants or inert. The latter substances are recycled to the reactor. To avoid buildup of the inert in the system, a purge stream is withdrawn from the recycle. The feed to the reactor not the fresh feed to the process contains molmol and mol inert. The singlepass conversion of hydrogen is Calculate the molar flow rates and molar compositions of the fresh feed, the total feed to the reactor, the recycle stream, and the purge stream for a production rate of molCH
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


