Question
microscale (100 mg solute mixture in 2 mL organic solvent) Your task is to provide a procedure for purifying a desired organic compound by removing
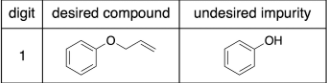
microscale (100 mg solute mixture in 2 mL organic solvent)
Your task is to provide a procedure for purifying a desired organic compound by removing an undesired impurity, using the technique of extraction.
To see what mixture you have been assigned, and whether you are working on regular scale (macroscale) or microscale, refer to this Google Doc (Links to an external site.). The scale will influence how you go about performing the isolation of the desired compound.
The scenario starts with a solution of the desired compound plus undesired impurity in the immiscible organic solvent of your choice. The solvent choice is not critical. However, if you take its density into consideration, your choice of solvent could make the procedure shorter.
For reagents, you can assume that you have the following at your disposal:
- 5% aqueous HCl
- 5% aqueous NaOH
- saturated sodium bicarbonate
- saturated sodium carbonate
- saturated aqueous sodium chloride
- sodium sulfate
- extra immiscible organic solvent of your choice (e.g. if your initial mixture is dissolved in diethyl ether, extra diethyl ether will be available)
Any laboratory glassware and equipment needed for the procedure (e.g. separatory funnel; rotary evaporator) would be available as well.
Provide a written procedure for purifying the desired compound, starting from the impure organic solution and ending with the pure desired compound. The procedure should be detailed enough that an undergraduate student that has successfully completed the "live" CHEM325 lab could follow your procedure successfully and safely. Some guidance:
- They would need to be told what steps to perform, but it will be understood that they know how to perform the techniques involved. For example, "extract solution X with solution Y" doesn't require explaining how to grip a separatory funnel.
- They would need to be told whether the organic layer is the upper or the lower layer.
- They also need to know what hazards are involved and what precautions to take. However, for this exercise, assume that the SDSs for all the compounds and reagents have already been reviewed with the student--you don't have to look up SDSs for this exercise.
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


