Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Module 3 1. Reaction between Brand H2O2 in acidic medium is as follows 2Br + H2O2 + 2H+ Brz+ 2H20 The proposed mechanism is K.
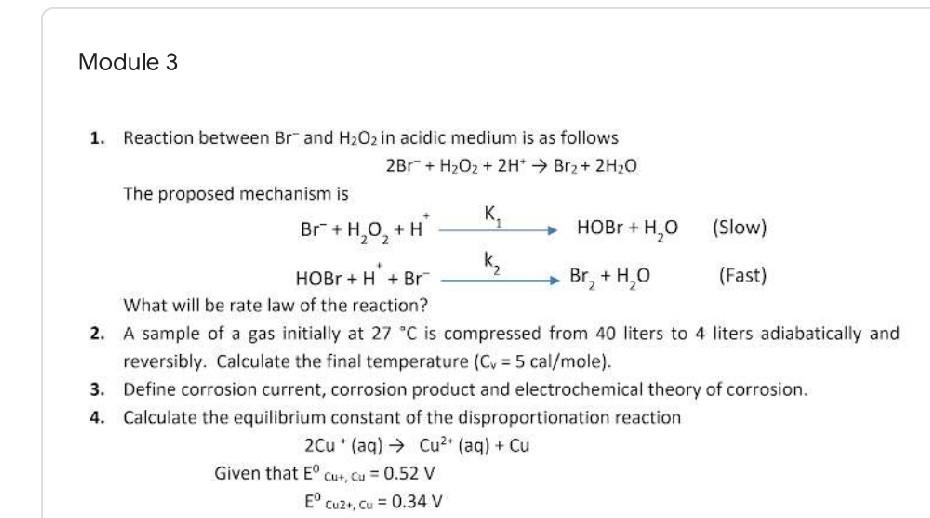
Module 3 1. Reaction between Brand H2O2 in acidic medium is as follows 2Br + H2O2 + 2H+ Brz+ 2H20 The proposed mechanism is K. Br +H,0,+H HOBr+H,0 (Slow) HOBr + H + Br k, Br, +HO (Fast) What will be rate law of the reaction? 2. A sample of a gas initially at 27 C is compressed from 40 liters to 4 liters adiabatically and reversibly. Calculate the final temperature (Cv = 5 cal/mole). 3. Define corrosion current, corrosion product and electrochemical theory of corrosion. 4. Calculate the equilibrium constant of the disproportionation reaction 2Cu' (aq) Cu? (aq) + Cu Given that ECut, cu = 0.52 V E' Cuz, cu = 0.34 V
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


